If you want to work with amorphous materials, like polymers and glasses, you’ll need to know about glass transition temperature. This information tells you which material keeps or loses its properties in higher temperatures, so is a must when choosing materials. This article will explain exactly what it is and how it works, as well as why it’s an important part of the 3D printing process.
What Is Glass Transition Temperature?
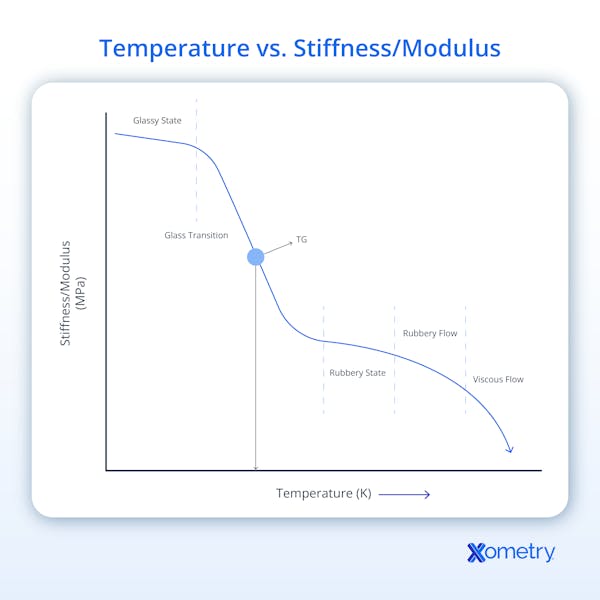
The point at which an amorphous material transitions from a hard and brittle state to a softer, more flexible one—i.e., when its polymer chains have been made more flexible with higher temperatures—is its glass transition temperature (Tg). This is sometimes also called the critical temperature and can predict material behavior in all temperatures and during processing, like extrusion or molding. If the material is kept at temperatures less than its Tg, it will stay hard and glass-like, but if subjected to temperatures higher than that, it will get viscous or rubbery.
To get a little more technical, at its Tg, a material’s thermal energy overpowers its intermolecular forces, and this allows the polymer chains to move more freely. The change is gradual and happens over a temperature range (unlike melting). It can be measured in Celsius (°C), Fahrenheit, or Kelvin. This transition will change aspects of the material, like its stiffness, ductility, conductivity, stability, durability, longevity, creep deformation resistance, and thermal, electrical, and mechanical properties.
What Are the Units Used for Glass Transition Temperature?
Glass transition temperature can be given in degrees Celsius (°C), degrees Fahrenheit (°F), and Kelvin (K). This temperature represents the point at which heating or cooling causes an amorphous material to change between a hard, glassy state and a rubbery, viscous state.
How Does the Glass Transition Temperature Work?
At the glass transition temperature, the energy contained in the polymer chains begins to match or exceed the energy contained in their intermolecular bonds. This results in an increase in molecular mobility and a gradual shift from a rigid to a more flexible state. Below Tg, the substance is in a glassy state with a highly ordered, rigid, and brittle structure. The material's flexibility and molecular mobility increase once Tg is reached, enabling it to deform under stress or flow under shear. The transition is dictated by internal energy within the molecular chains; when they become more mobile, they alter the polymer’s overall physical characteristics.
What is the Importance of Glass Transition Temperature?
Tg is significant because of how it affects the mechanical, thermal, and processing characteristics of polymers. The Tg value, for instance, can determine the temperature range over which a polymer will be considered stable and retain its mechanical properties. A polymer will be hard and brittle below Tg and soft and deformable above Tg. Tg has an impact on polymer processing as well because it specifies the range of temperatures where the item’s shape can be altered without major changes to its overall form.The glass transition temperature can also have an impact on a product's shelf life since polymers can deteriorate or change shape if stored at temperatures above Tg.
What Is the Importance of Tg in 3D Printing?
In 3D printing, Tg directly impacts material performance, print quality, and how stiff, tough, and dimensionally stable the final product will be. It will dictate the best temperature for extrusion, melting, and fusion of layers. If the material isn’t kept within a stable temperature range, the layers could crack or bond incorrectly (if too cool) or warp, deform, or collapse under their own weight (if too hot). Take PLA as an example; its relatively low Tg (approx. 55–60°C) makes it easy to print with but prone to softening and deformation even at moderate temperatures. ABS, though, has better resistance to warping and deformation during printing since its Tg is around 105°C.
It’s not the only value you need to know to work out the stability and durability of a 3D printed part—this depends on other things like material properties, printing parameters, and design considerations. To predict how it will behave, various techniques and tools can be used, like finite element analysis (FEA). This simulates the mechanical behavior of a 3D printed part under different conditions, including static and dynamic loads, temperature and humidity changes, and various environments. In this way, you can gain insights into stress and strain distribution within the part, find potential failure modes, and optimize your design and material selection accordingly.
How Does the Tg of 3D Printing Material Affect Its Printability?
The range of temperatures at which a 3D printing material can be extruded, melted, and fused into layers depends on its Tg (glass transition temperature). The material will not flow properly if the temperature is too low, producing prints that are brittle and weak. If it is too high, the material will deteriorate and produce surfaces with poor adhesion.
How Does the 3D Printing Cooling Rate Impact Tg?
The cooling rate during 3D printing can significantly influence the glass transition temperature of printed parts. Tg tends to rise with slower cooling rates and fall with faster cooling rates. This is because slower cooling rates give molecular chains more time to align and create stronger bonds, increasing Tg. If the material cools quickly, on the other hand, the molecules don’t have time to reach that optimal alignment, so they have weaker bonds. Depending on the specific printing material and printing conditions, the precise effect of the cooling rate on Tg can change.
Can Tg Predict 3D Printed Part Stability and Durability?
No, Tg cannot predict a 3D-printed part’s stability and durability, although it is a significant material property. Other properties, plus printing parameters and design considerations also influence the printed item’s performance. However, there are techniques and tools that can help you predict the strength and resilience of 3D-printed parts. For instance, finite element analysis (FEA) can simulate the mechanical behavior of a 3D printed part under a variety of conditions such as static and dynamic loads, temperature and humidity changes, and exposure to various environments. You can get insights into the stress and strain distribution within the part, identify potential failure modes, and optimize the design and material selection for improved stability and durability by analyzing the FEA results.
Is It Possible To Change a 3D Printing Material's Tg Chemically?
Yes, by changing the chemistry, you can alter the Tg of a material used for 3D printing. A change in its chemical makeup or the simple addition of chemical agents will change its properties. For example, you can increase a PLA print’s glass transition temperature by adding additives, but doing so may also compromise its strength and infusibility.
What Are the Different Methods of Measuring Glass Transition Temperature?
To work out a material’s Tg, there are several differential scanning calorimetry and dynamic mechanical analysis methods available. These are listed in the table below.
| Method | What It Measures | How It Measures | Best For |
|---|---|---|---|
Method Differential Scanning Calorimetry (DSC) | What It Measures Heat flow associated with the material’s transition | How It Measures Provides data on the energy absorbed or released during the transition | Best For Materials with thermal transitions, i.e., polymers used in thermal applications |
Method Dynamic Mechanical Analysis (DMA) | What It Measures Changes in mechanical properties (i.e., stiffness) | How It Measures Deforms the polymer sinusoidally | Best For Materials that need mechanical properties, like composites or structural polymers |
Method Thermomechanical Analysis (TMA) | What It Measures Dimensional changes during heating | How It Measures Calculates from the material’s plot of thermal expansion/ compression vs. the temperature | Best For Measuring thermal expansion and contraction in things like coatings or adhesives |
Method Dielectric Analysis (DEA) | What It Measures Changes in electrical properties during transition | How It Measures Monitors changes in dielectric constant or loss factor | Best For Polymers used for electronics or insulation |
Method Fourier Transform Infrared Spectroscopy (FTIR) | What It Measures Changes in molecular vibrations near Tg | How It Measures Detects changes in infrared absorbance corresponding to molecular bond vibrations | Best For Chemically analyzing changes to molecular structure in blends and copolymers |
Method Dynamic vapor sorption (DVS) | What It Measures Changes in the material’s ability to absorb water vapor | How It Measures Monitors weight changes due to vapor absorption | Best For Hydrophilic materials, finding environmental effects on polymers, like packaging films |
Glass Transition Temperature Measuring Methods
| Polymer Type | Example | Approximate Tg in °C | Measuring Method |
|---|---|---|---|
Polymer Type Crystalline* | Example Polypropylene (PP) | Approximate Tg in °C 165°C (melting temp) | Measuring Method DSC, TMA, DMA |
Polymer Type Crystalline* | Example Polyethylene terephthalate (PET) | Approximate Tg in °C 250°C (melting temp) | Measuring Method DSC, DMA |
Polymer Type Crystalline* | Example Nylon (polyamide, PA) | Approximate Tg in °C 220°C (melting temp) | Measuring Method DSC, DMA |
Polymer Type Semi-crystalline** | Example Polyethylene (PE) | Approximate Tg in °C -100°C | Measuring Method DSC, DMA |
Polymer Type Semi-crystalline** | Example Polyvinylidene fluoride (PVDF) | Approximate Tg in °C -35°C | Measuring Method DSC, DMA |
Polymer Type Semi-crystalline** | Example Polyethylene terephthalate (PET) | Approximate Tg in °C 70°C | Measuring Method DSC, DMA |
Polymer Type Amorphous*** | Example Polystyrene (PS) | Approximate Tg in °C 100°C | Measuring Method DMA, TMA, DSC |
Polymer Type Amorphous*** | Example Polycarbonate (PC) | Approximate Tg in °C 150°C | Measuring Method DSC, DMA |
Polymer Type Amorphous*** | Example Polymethyl methacrylate (PMMA) | Approximate Tg in °C 105°C | Measuring Method DSC, DMA |
Glass Transition Materials and Their Properties
* these have no clear Tg because they don’t have amorphous regions; melting temp is used to measure transition
** have both amorphous and crystalline regions; the Tg usually refers to the amorphous part
*** no crystalline regions, so have very distinct Tg
1. Crystalline Polymers
Since crystalline polymers do not have clearly defined Tg, these measurements do not apply to them. Crystalline polymers are characterized by other properties instead.
2. Semi-Crystalline Polymers
Semi-crystalline polymers are substances that contain both disordered amorphous and ordered crystalline regions. Differential scanning calorimetry (DSC) or dynamic mechanical analysis (DMA) is used to measure the glass transition temperature of such polymers. The semi-crystalline polymer polyethylene, with a Tg of about -100°C, is one common example.
3. Amorphous Polymers
Amorphous polymeric materials change from a hard, brittle state to a rubbery, flexible state at a temperature known as the glass transition temperature. Dynamic mechanical analysis (DMA), thermomechanical analysis (TMA), differential scanning calorimetry (DSC), and other methods can be employed to identify this value. In polystyrene, for instance, that temperature happens to be about 100 °C.

What Are the Different Factors That Affect the Tg Value of a Material?
Material selection is Tg information comes in handy so you can make sure it can do what you want it to do. For instance, choosing polystyrene, which has a Tg of approximately 100°C, to make ovenware is a disaster waiting to happen. That’s a very generic example, but you get the idea. The Tg will also let you know how mechanically stable and thermally resistant a material is, as well as its expected shelf life (lower Tg options typically have a shorter life because they’re more prone to structural changes) and the best manufacturing methods to process.
The measurement only works with amorphous materials (not metals and ceramics) and provides only one aspect of the material’s overall properties (important nonetheless). Also, it might not be a reliable predictor of a material’s behavior because measurement sensitivities can lead to report variations/inconsistencies, and the value can change depending on several factors—this will then affect how the material reacts in cooling and hardening (i.e., if its properties change), and ultimately how it can be processed. These include:
1. Pressure
High pressure compresses the polymer chains and increases the rigidity.
2. Moisture
This acts as a plasticizer, so will lower the Tg as it disrupts the molecular bonds and increases chain flexibility.
3. Composition
The Tg of a material made from two different copolymers will be a mix of both the materials’ Tg values, and additives—like fillers and stabilizers—will also likely affect the Tg.
4. Molecular Weight
If this is higher in a material, then its Tg will be higher, too, due to the stronger intermolecular forces.
5. Chemical Structure
Flexible polymer backbones have lower Tg, while rigid structures have a higher one, and cross-linking can restrict molecular motion to make it even more rigid.
6. Plasticizers
These will reduce a Tg in a material by increasing the free volume between the polymer chains, which increases mobility.
7. Crystallinity
Highly crystalline polymers tend to have higher Tgs because their more ordered structure reduces the chain mobility.
8. Polar Groups
Adding these strengthens the intermolecular forces and will raise the value.
9. Environmental Factors
Extremes temperatures, UV radiation, or exposure to solvents or acids can degrade molecular bonds or alter polymer structure over time.
10. Thermal History
Rapid cooling can trap the polymer in a less stable, higher-energy state (lowering Tg), while annealing could make it more stable (increasing Tg).
The glass transition temperature is, generally, the temperature at which a material transitions from pliable to solid. This will effect how warping forces and internal stresses manifest, as well as how much a part is going to want to move around during the 3D printing process.Christian Tsu-RaunTeam Lead, Manual Quoting
How Does Tg Impact the Durability and Longevity of Polymer Products?
Tg has an impact on the toughness and longevity of polymer products. Polymers with higher Tg values tend to last longer because they are more resistant to creep deformation and have better mechanical properties at higher temperatures.
What Are the Advantages of Tg?
The glass transition temperature (Tg) is an integral property of amorphous materials. There are several reasons you’d want to know it:
- Predicting the Mechanical Properties: A material's properties can be predicted based on temperature. The material is rubbery and flexible above Tg but stiff and brittle below Tg.
- Stability: Tg also indicates the thermal stability of the material. Higher-Tg materials can withstand higher temperatures before experiencing significant structural changes and are more resistant to thermal degradation.
- Processing: Tg is used to identify the ideal processing temperature range to prevent structural changes in the material and maintain the desired properties.
- Shelf-life: The Tg can also be used to estimate a material's shelf life. Lower-Tg materials are more susceptible to structural changes over time, which results in shorter shelf life.
What Are the Disadvantages of Tg?
Materials with a glass transition temperature have many beneficial uses in materials science and engineering, but they also have their own drawbacks. Here are a few examples:
- Limited Applicability: The Tg concept only applies to materials that experience a glass transition cannot be used to explain how substances like metals and ceramics behave.
- Sensitivity to Sample Preparation: A material's thermal history and preparation can have an impact on its Tg. It can be challenging to compare data from different sources because different Tg measurement techniques can produce inconsistent results.
- Dependence on Composition: The overall Tg can depend on the molecular weight of a polymer or the number of additives in a glass. This implies that Tg might not always be a reliable predictor of a material's behavior.
- Not a Complete Characterization: Tg is just one of many attributes that characterize a material's behavior. It does not give a comprehensive picture of its mechanical or thermal characteristics.
Frequently Asked Questions About Glass Transition Temperature
Is Tg a Stiffness Indicator for a Material?
Although a material’s Tg is linked to stiffness, it’s not a direct indicator of it because stiffness depends on other factors as well, such as degree of crystallinity, cross-linking density, and any fillers/additives that have been included. Also, unlike Tg, stiffness is a measure of a material's resistance to deformation under stress at a specific temperature.
Is Tg a Temperature at Which a Material Changes From a Solid to a Liquid State?
No, the temperature at which a substance transforms from a solid to a liquid state is not the same as the Tg. Rather, Tg is the temperature at which a material changes from a glassy state to a rubbery state, which is indicated by a change in mechanical properties like stiffness and viscosity.
Does Tg Have an Impact on How Polymers Are Manufactured and Processed?
Yes, the Tg has a significant effect on the production and processing of polymers. The Tg establishes a polymer's processing temperature range and affects how the substance behaves during cooling and solidification, which in turn affects its mechanical and physical characteristics. Various processing techniques and environmental factors can also affect the Tg of a polymer.
Can a Material's Glass Transition Temperature Be Changed as It Is Being Synthesized?
Yes, changing the chemical makeup or processing conditions during the synthesis process will change a material's Tg. For example, if you add a plasticizer, this can lower the value, but if you increase the cross-linking, it’ll get higher.
What Is the Difference Between Glass Transition Temperature and Melting Temperature?
A material’s melting temperature (Tm) is the point at which it changes from a solid to a liquid state (the way a material behaves when flexible isn’t the same as when it’s liquid). The Tg, as we’ve seen, is when the material’s state reverses from hard and brittle to rubbery and flexible.
How Xometry Can Help
To learn more about glass transition temperature, or any other related concept, you can reach out to one of our representatives. Xometry provides lots of services for all your manufacturing, prototyping and production needs. Visit our website to learn more, or upload your designs to the Xometry Instant Quoting Engine® and get started straight away.
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.


