Polymers are materials composed of repeating chains of individual atoms or molecules. Polymers can be naturally occurring (such as cellulose, latex, and rubber) or synthetic (like nylon, polyethylene, and polypropylene). The terms “polymer” and “plastic” are often used interchangeably, but there are noticeable differences between the two. Plastics are hydrocarbon-based polymeric materials derived from crude oil and natural gas and are a subset of polymers. Therefore, while all plastics are considered polymers, not all polymers are considered plastics. Differences between polymers vs. plastic also exist concerning recyclability, flexibility, and strength. This article will define polymers and plastics and discuss the differences between the two types of material.
What Is Polymer?
A polymer is a natural or synthetic chemical compound that is composed of repeating strings of large, chemically-bonded molecules, or monomers. Polymers can be characterized as homopolymers (polymers comprised of one type of monomer) or as copolymers (polymers comprised of two or more monomers). Polymers constitute the basis of many materials in the universe, from proteins and nucleic acids in organisms to minerals like quartz and diamond, to synthetic materials like plastics, paper, and rubbers.
What Is Polymer Made Of?
A polymer is made of chemically-bonded chains of molecules or monomers. These monomers can be simple and comprised of a few atoms, or be complex functional groups of atoms. The chemical composition and size of the individual monomers determine how the polymer interacts with itself and its surrounding environment.
What Are the Properties of Polymer?
The properties of polymers differ based on their chemical composition. However, the general properties of polymers are listed below:
- With longer chain lengths and more cross-linked bonds polymers tend to have higher tensile strength.
- With longer chains and higher intermolecular forces, they have higher melting and boiling points.
- They are resistant to chemicals due to their low reactivity.
- They are formed by hydrogen and ionic bonds.
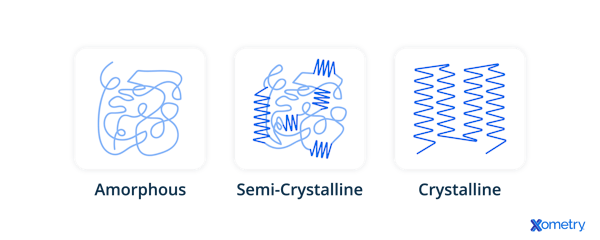
- The crystal structure of polymers is typically semi-crystalline or amorphous.
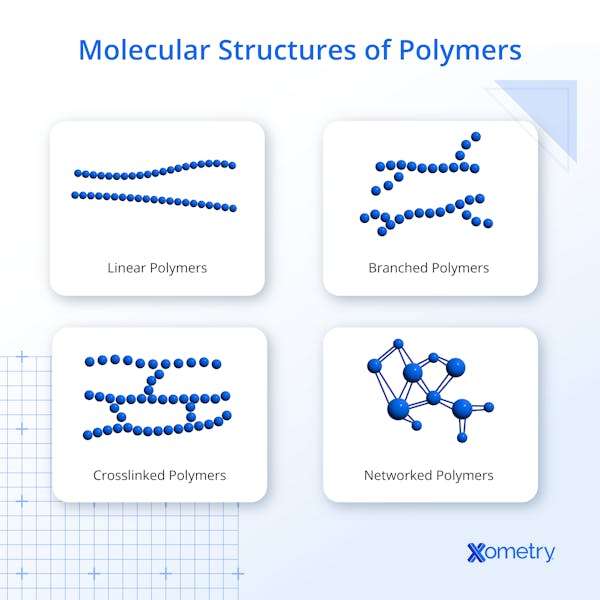
What Is the Structure of Polymer?
There are three structures of polymers: linear, branched, and network or cross-linked. Linear polymers consist of a chain of monomers, typically called the backbone in branched and cross-linked polymers. Bonded to the backbone are other elements or compounds, most frequently hydrogen, fluoride, chlorine, boron, and iodine. The additional bonded elements or compounds called the pendant groups, hang from the chain of carbon atoms like charms on a bracelet. This is a branched polymer. The pendant groups can connect, or cross-link, to other parallel chains of molecules depending on the number of valence electrons available in these pendant groups. These are considered network polymers. Shorter cross-links can strengthen a polymer while longer cross-links can make a polymer more flexible.
What Is the Advantage of Polymer Compared to Plastic?
There are many advantages of using polymer compared to plastic. The list below describes some of the advantages:
- Natural polymers are not derived from crude oil.
- Natural polymers do not release toxic fumes when burned.
- Polymers are generally more sustainable than plastics since they are more biodegradable than synthetic plastics.
What Are Examples of Polymers?
Polymers are found everywhere. They are inside each living organism and can be found in rocks and minerals. Some examples of polymers are listed below:
- Cellulose and lignin are natural polymers that form the basis for plants and trees.
- Nylon, polypropylene, and polyethylene are synthetic polymers that are classified as thermoplastics.
- Elastomeric materials like rubber and latex.
What Is Plastic?
Plastics are synthetic polymeric materials derived from petroleum. Plastics are produced through a polymerization or polycondensation process. In these processes, oil and natural gas are refined to form gases like ethane and propane. The produced ethane and propane are then heated to form monomers like ethylene and propylene. The monomers and a catalyst are mixed to form a polymer. This mixture is then extruded, cooled, and cut into pellets which are then shipped to plastics fabrication companies around the world.
What Is Plastic Made Of?
Plastic is made from organic raw materials, primarily crude oil, but it can also be made from coal, natural gas, and cellulose. Oil is a hydrocarbon material that, when refined, is split into different materials, or fractions, that differ based on their molecules’ size and structure. Naphtha, one of the byproducts of the oil refining process, is the substance primarily used for plastics production.
What Are the Properties of Plastic?
There are two types of plastics: thermoplastics and thermosets. The properties of these two types of plastics differ depending on the particular thermoplastic or thermoset. However, a list of general properties of plastics is shown below:
- Thermoplastics melt when heated above their melting point and can be cooled and reheated several times.
- Thermosets are plastics that remain solid after curing and do not melt, even at high temperatures.
- Plastics are chemically stable.
- Plastics have low electrical and thermal conductivity.
- Plastics have a high strength-to-weight ratio.
What Is the Structure of Plastic?
Because plastics are considered polymers, plastics exhibit the same structures as polymers. Plastics can be linear polymers (like PVC), branched polymers (like low-density polyethylene), or cross-linked polymers (like bakelite and melamine). The chemical composition of the monomers that form the plastic determines its structure and type of polymer. Plastics can either be amorphous, in which case the polymer chains are not organized in a particular fashion, or semi-crystalline, where the polymer chains are ordered and mixed in with amorphous regions.
What Is the Advantage of Plastic Compared to Polymer?
There are many advantages of using plastic compared to a polymer. The list below describes some of the advantages:
- Production cost is low.
- Large amounts of plastic products can be made quickly.
- Plastic has a high strength-to-weight ratio.
- Plastic is a versatile material and can be used for a wide range of applications including use in consumer products, and industries such as: automotive, medical, aerospace, and heavy machinery.
What Are Examples of Plastics?
Some examples of plastics are listed below:
- Nylon
- Low-density polyethylene (LDPE)
- PVC
- Polyoxymethylene (POM)
- Polypropylene (PP)
Which Is Better for 3D Printing, Polymer or Plastic?
To say whether polymer or plastic is better for 3D printing largely depends on the application of the 3D-printed part. Polylactic acid (PLA) is a popular biodegradable polymer used for 3D printing of short life-cycle, non-load-bearing parts, like hobbyist prints. Whereas ABS, a thermoplastic polymer lauded for its strength, durability, and impact resistance, is often used to 3D-print toys, electronic enclosures, and automotive parts. The 3D-printing process for polymer vs. plastic will differ. Some polymers may be more difficult to print than plastics and vice versa.
What Are the Advantages of Using Polymer in 3D Printing?
The advantages of using polymers in 3D printing are listed below:
- Natural polymers like gelatin and alginate can be used to 3D print cell-loading hydrogels for human tissue and organ formation.
- Natural polymers are used as fillers in plastic 3D-printing filaments to reduce adverse environmental impact and improve biocompatibility.
What Are the Advantages of Using Plastic in 3D Printing?
The advantages of using plastic in 3D printing are listed below:
- 3D printing with plastics can produce strong, lightweight parts.
- 3D-printing plastic parts can replace some parts that are typically made from metals.
- 3D-printing plastic is an excellent method for proof of concept and prototyping new designs.
What Are the Disadvantages of Using Polymer in 3D Printing?
One disadvantage of using polymers in 3D printing is that a limited number of polymers are available for 3D printing.
What Are the Disadvantages of Using Plastic in 3D Printing?
The disadvantages of using plastic in 3D printing are listed below:
- A limited number of plastics are available for 3D printing.
- Many plastics release harmful fumes when 3D printing.
- 3D printing with non-biodegradable plastics can harm the environment since many 3D-printed objects are single-use or for proof of concept.
Why Is Polymer Better Than Plastic?
Polymer is better than plastic because many examples are more flexible, sustainable, and biocompatible. For instance, polymers derived from natural sources like lignin, cellulose, silk, and cotton, are often used in clothing, textiles, coatings, and adhesives due to their flexibility. Because these materials are naturally occurring, they break down more quickly in nature than synthetic plastics. Additionally, some polymers, like polyurethane and polyethylene glycol, are used in drug delivery systems and medical implants due to their biocompatibility. Plastics are not without their benefits, however. Ultimately, the determination of which is better, polymer vs. plastic, comes down to the application and volume of parts needed.
Are Polymers Stronger Than Plastic?
No, polymers are not stronger than plastic. However, the answer largely depends on the items being compared.
Are Polymers Considered Plastic?
Yes, all polymers derived from petroleum, such as thermoplastics and thermosets, are considered plastics. However, naturally occurring polymers such as cellulose, proteins, and cotton are not plastics.
Are All Polymers Plastics?
No, all polymers are not plastics. However, all plastics are considered polymers because all plastics contain repeating units of chains of molecules derived from petroleum.
What Polymers Are Not Plastic?
Polymers that are not petrochemical-based are not considered plastics. Examples include: wool, paper, wood (cellulose), silk, and natural rubber.
What Is the Difference Between Polymers and Plastics in Terms of Tensile Strength?
The tensile strength of polymer vs. plastic largely depends on the crystal structure of the items being compared. Tensile strength refers to the maximum strength a material can sustain before fracture when stretched. The material’s crystal structure has a large influence on its tensile strength. Materials with compact crystal structures with more slip systems are generally more ductile and have higher tensile strength.
What Is the Difference Between Polymers and Plastics in Terms of Hardness?
Hardness is a material’s ability to resist localized deformation. When comparing the hardness of polymers vs. plastics, plastics tend to be harder than polymers since many plastics are cross-linked copolymers composed of different monomers. The cross-linked structure makes plastics more resistant to localized deformation. For more information, see our guide on Hardness.
What Is the Difference Between Polymers and Plastics in Terms of Impact Strength?
Impact strength is a material’s ability to resist fracture due to a sudden applied load or shock. The impact strength of polymers vs. plastic largely depends on the material’s crystal structure. Semi-crystalline plastics tend to have higher impact strength than polymers since their close-packed crystal structures are harder and more resistant to deformation.
Summary
This article presented polymers vs. plastic, explained what each is, and discussed the various differences between the two of them. To learn more about polymers vs. plastic, contact a Xometry representative.
Xometry provides a wide range of manufacturing capabilities and other value-added services for all of your prototyping and production needs. Visit our website to learn more or to request a free, no-obligation quote.
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.

