The ultra-elastic material nylon has comfortably found its place across industries, whether it’s molded into a washer for an airplane or found within the ingredient list of a skincare product. It’s abundant and easy to produce, but not all nylons are exactly the same. They can be broken down into several types based on their amide group, and because of some differences, can be separated out for use in certain applications and manufacturing processes, which could be anything from CNC machining to 3D printing.
Two types you may be familiar with are nylon 6 and nylon 6/12, which have some of the same properties, but also have a few differences. We’ll explain how both are made and what factors make them distinct from one another. You’ll also learn what applications you can incorporate nylon into, some of which are common everyday products and others are more niche items.
What Is Nylon 6?
Nylon 6 is the same thing as polycaprolactam—which admittedly doesn’t roll off the tongue as easily. It’s a polymer that’s created through the polymerization of caprolactam, which is its biggest building block.
The pipeline from its invention, courtesy of German chemist Paul Schlack, to its use in manufacturing and commerce was a quick on. Even to this day, it’s a popular material that’s still being improved upon. There are seven other types of nylon out there, but the easiest way to remember this specific kind and its structure is through its name, six, which is because it holds six carbon atoms.
How Is Nylon 6 Produced?
Nylon 6 is produced by ring-opening polymerization of caprolactam. Nylon 6 is primarily made by heating caprolactam to 250 degrees C and adding around 4% water. A carbonyl group is present in caprolactam and when water molecules are present, the oxygen of that carbonyl group absorbs a proton from the water molecule. From this, the oxygen atom has a positive electrical charge. Since this is undesirable, the molecule rearranges and leaves the positive charge on the carbonyl carbon atom. The nucleophile, in this case OH–, can then attack this carbon atom and as a consequence, the caprolactam molecule's ring is opened. This opened segment can force another ring to open, resulting in ring-opening polymerization. The amide bond within each caprolactam molecule is broken during polymerization. The active groups on both sides generate two new bonds as the monomer becomes part of the polymer backbone.
Properties of Nylon 6
There are a handful of characteristics that make nylon 6 stand out from its fellow nylons and other materials used in similar capacities. To start, it’s extremely elastic and has an impressive tensile strength, too. It’s capable of handling exposure to alkali and acidic substances and it has resistance to different types of abrasion. It can handle up to 220℃ before it melts, but its glass transition temperature sits at 48℃. If you look at a nylon 6 filament, you’ll notice its smoothness and featureless appearance is similar to glass. It’s also impressive at holding water (managing to absorb around 2.4%). Altogether, these properties make it a worthy candidate for automotive, aerospace, cosmetic, and consumer products, and it can serve as a technical nutrient.
It’s easier to visualize nylon 6’s chemical formula and molecular structure by taking a look at the image below.
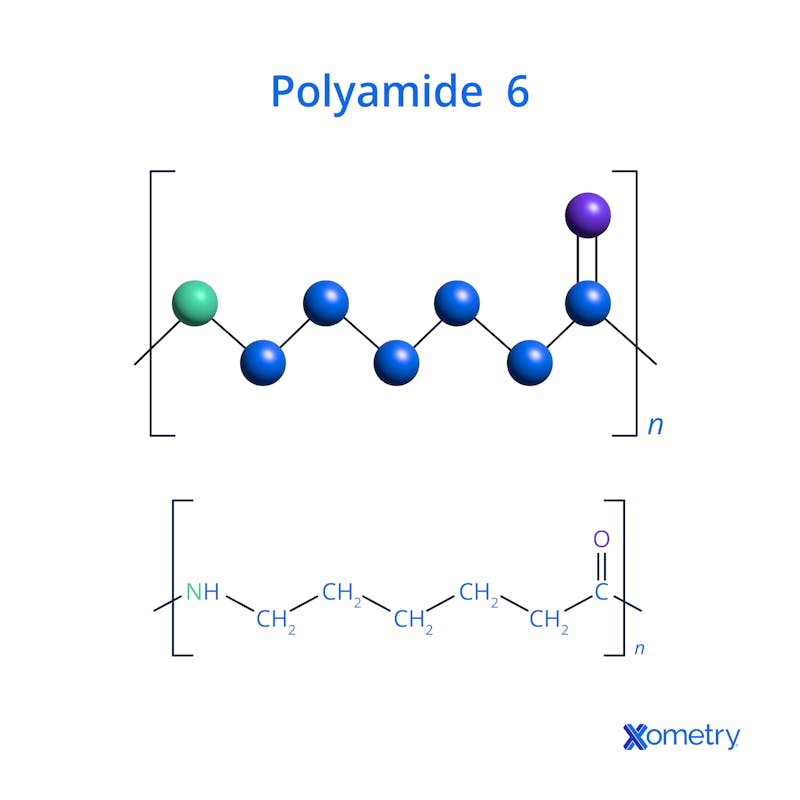
What Are the Uses of Nylon 6?
Nylon 6 is a popular choice for applications where its durability, toughness, and resistance to wear and tear will come in handy, like the following:
- Fibers
- Toothbrushes
- Guitar strings and picks
- Gears
- Panel catches
- Circuit insulation boards
- Power tool housing
- Medical implants
- Films, wraps, and packaging
What Are the Advantages of Nylon 6?
The following advantages make nylon 6 a great choice for certain applications:
- Nylon 6 provides excellent stiffness and abrasion resistance
- This material is perfectly fine for use in injection molding
- It’s a great material if you need objects that are resistant to impact and wear and tear
- It has serious elasticity, and even better, it can return to its original shape even after distorting or being subject to tension or pressure
- This type of nylon is a great option if you’re interested in dying it different colors because it holds and shows dye well
What Are the Disadvantages of Nylon 6?
On the reverse, there are also a few disadvantages to be aware of with this type of nylon:
- In comparison to other plastics and materials, nylon 6 has a low melting point (220℃)
- Although its water absorption might be a plus, it’s hygroscopic, so it’ll pull water from the air and its surroundings
- When it’s exposed to light and high temperatures, you’ll find that nylon 6 will lose its strength, toughness, and structural integrity
- It’s not UV resistant, and both UV rays and light can discolor it

Nylon 6 has higher tensile strength and stiffness, which make it a good choice for applications where rigidity and strength are critical. Projects that have an application that demands high strength, rigidity, and cost-efficiency in environments with minimal water or humidity exposure should choose Nylon 6. Nylon 6/12 has lower strength and stiffness compared to Nylon 6. However, Nylon 6/12 has better flexibility and impact resistance. Projects that have an application requiring superior moisture resistance, dimensional stability, and flexibility — especially in challenging environments like marine or humid conditions — should choose Nylon 6/12.Kurt PokopacSolutions Engineer
How Does Nylon 6 Affect Human Health?
Nylon 6 is entirely free of all toxic properties and does not irritate the skin. This material is chemically inert and is proclaimed a non-dangerous substance by the GHS (Globally Harmonized System of Classification and Labelling of Chemicals). However, nylon 6 may be harmful if accidentally inhaled, since the particles are bio-persistent and will likely remain in the lungs for an extended period, causing respiratory tract irritation. Moreover, this polymer may cause skin irritation if absorbed through the skin. If molten material comes in contact with skin, the person should not attempt to remove it since doing so might cause severe tissue damage. Accidental indigestion of nylon 6 might also cause health problems, such as gastrointestinal blockage.
Can Nylon 6 Be Used in Plastic Injection Molding?
Yes, Nylon 6 is a suitable material for injection molding. The resulting molded nylon parts possess great strength, as well as chemical and temperature resistance. When molding nylon 6, the material is sometimes injected with a specified amount of glass fibers (usually between 10% and 40%) to boost its tensile strength. Glass fibers improve stiffness while also influencing how a part behaves under different conditions. For example, when subjected to stress, an item that is molded by a mixture of nylon and glass would break more quickly and easily than an item that has been molded with only nylon. Moreover, since UV radiation can be harmful to nylon, a UV stabilizer is frequently added to the material before injection molding to decrease the possible degradation of the item with time.
What Is Nylon 6/12?
Cousin to nylon 6, nylon 6/12 is also a thermoplastic, but it was created to solve for nylon 6’s moisture absorption—which has proven to be problematic for some applications. It also goes by the name of polyamide 6/12 or PA 612, and is a mix of two different compounds, which gives it its double-number name. The molecular formula is C18H36N2O3, which has quite a few more atoms of each element in place than its precursor.
How Is Nylon 6/12 Produced?
Nylon 6/12 is produced by combining two compounds in one precursor, just like all other nylon polymers with a two-number nomenclature. This precursor is then polymerized to create the final material. The numbers after the word nylon represent the number of carbon atoms in each initial reactant. Nylon 6/12 is made up of a diamine known as hexamethylenediamine and a diacid known as dodecanedioic acid. This acid contains 12 carbons, which increases the distance between the amide groups, resulting in worse mechanical characteristics and a lower melting point. However, because the amide group is responsible for the polymer's moisture affinity, nylon 6/12 is more resistant to moisture absorption than other types of nylon.
Properties of Nylon 6/12
To create nylon 6/12, you must polymerize nylon 6 and add in both hexamethylenediamine and dodecanedioic acid, which stretch out the distance between the amide groups. In terms of nylon properties, the result is a material that’s not as strong and has less resistance to chemicals. Its melting point is just a smidge lower than nylon 6, at 218 ℃—for a higher melting point than both, look to nylon 66. That said, it’s a better option if moisture resistance is more important and you need a material that won’t lap up water from the surrounding air.
What Are the Uses of Nylon 6/12?
Nylon 6/12 is usually a popular material for extruded tubes and several other uses, like the following:
- Electric appliances
- Personal care products, cosmetics, and skincare
- Parts for cars and other vehicles
- Brush bristles
- Fuel systems
What Are the Advantages of Nylon 6/12?
These are the main advantages to working with nylon 6/12:
- This type of nylon absorbs much less than its cousin nylon 6, and therefore is less prone to swelling
- It also has great resistance to wear and tear and has a decent level of strength, too
- When you compare it to other similar polyamides, 6/12 provides better electrical insulation and has a lower chance of cracking under stress
- While it’s not as resistant as nylon 6, 6/12 is capable of handling certain levels of heat, and it’s smoother than other polymers
- It’s also a suitable option for injection molding because of its ability to be melted down (without deteriorating) and then cooled and molded
What Are the Disadvantages of Nylon 6/12?
There are also several disadvantages that this type of nylon brings with it:
- Nylon 6/12 has much weaker resistance when it comes into contact with acidic and alkaline solutions
- It’s not as widely available as nylon 6, and, because of its specific purpose, it tends to be more expensive than other types of nylon
- It doesn’t have high crystallinity and therefore also has a lower heat deflection temperature and tends to shrink more within molds than other plastics do
How Does Nylon 6/12 Affect Human Health?
Nylon 6/12 has been proven to be generally safe for human health. Since it is commonly used as bulking and opacifying agent in cosmetics, the Cosmetic Ingredient Review Expert Panel had to test the possible toxicity of the polymer. After examining relevant animal and human data, the panel stated that nylon 6/12 is not likely to penetrate the skin and as such is considered non-toxic. Inhaled droplets/particles deposited in the nasopharyngeal and thoracic areas of the respiratory system may have harmful consequences depending on their chemical and other features. However, the current evidence shows that unintentional inhalation would not lead to major local respiratory or systemic problems except for maybe irritation, given the minimal actual exposure in the breathing zone and the quantities at which the chemicals are employed. Furthermore, accidental indigestion of nylon 6/12 might result in inflammation and other health issues. When in contact with eyes or skin, nylon fibers may irritate the skin and cause eye discomfort. Aside from particle-induced mechanical irritation of the skin and eyes, no significant health consequences are indicated.
Why Is Nylon 6/12 So Poor at Absorbing Moisture?
Nylon 6/12 is poor at absorbing moisture because it was created to prevent moisture absorption. Nylon 6/12 is a copolymer, which means it's made up of 12 different di-acids with 12 carbon chains. When these di-acids are combined with a 6-carbon diamine, they polymerize into nylon. Nylon 6/12 has a higher number of carbon atoms separating the amine groups in its chemical structure than nylon 6, and this ratio and chemical geometry give nylon 6/12 greater resistance to moisture absorption.
Can Nylon 6/12 Be Used in Plastic Injection Molding?
Yes, Nylon 6/12 can be used in injection molding. At its melting point, nylon 6/12, like other thermoplastics, becomes liquid and can be heated, chilled, and then reheated without any significant deterioration. Because of this, PA 612 is a good material to be injection molded into various plastic items and then eventually recycled. Nylon 6/12 can be filled with glass, minerals, impact modifiers, flame retardants, UV stabilizers, and other modifiers to improve some of its properties. Injection molding glass-filled nylon, for example, boosts tensile strength and makes the designed material less flexible and stiffer by adding glass fibers to polyamide plastic. Other common additives can be used with nylon to generate toughened, conductive, and high-temperature polyamide plastics with increased strength, durability, and flexibility.
Comparing Nylon 6 and Nylon 6/12
Overall, you’ll find that nylon 6 offers better resistance to chemicals, acids, bases, and impact than its polymerized counterpart nylon 6/12. However, if water absorption is an issue for your particular application, you’ll want to lean on 6/12 for its reduced ability to soak up water that it comes into contact with—in the air or otherwise. Side by side, the two are relatively similar when it comes to strength. Despite nylon 6/12’s lack of resistance to chemicals and acids, it tends to be slightly more expensive because it’s not produced as frequently.
How Xometry Can Help
These types of nylon are popular in various sectors, and to help businesses with these needs, we have numerous services that use nylon 6 and nylon 6/12. These include 3D printing, compression molding, plastic extrusion, and custom plastic fabrication. You can get a free quote today for any of these through our website.
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry's network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.


