Polyethylene (PE) is a versatile, low-cost thermoplastic made from ethylene monomers. Polyethylene plastic is used across industries (packaging, construction, automotive, and medical), valued for its flexibility, durability, and chemical resistance. The material is found in products (bags, films, and containers) due to its lightweight strength. Polyethylene material is used in pipes and geomembranes for moisture and chemical resistance in construction. Medical-grade forms, such as ultra-high-molecular-weight polyethylene (UHMWPE), are utilized in tubing and prosthetics for their biocompatibility and resistance to sterilization. The main types of polyethylene include low-density polyethylene (LDPE), which is flexible and used in bags and films, and high-density polyethylene (HDPE), which is strong and rigid, used in containers and pipes. Linear low-density polyethylene (LLDPE) and ultra-high-molecular-weight polyethylene (UHMWPE) offer higher toughness and wear resistance. Each type’s molecular structure gives it unique properties, making polyethylene one of the most used plastics worldwide.
What is Polyethylene (PE)?
Polyethylene (PE) is a thermoplastic polymer made from ethylene monomers and is the most produced plastic in the world due to its cost-effectiveness, ease of processing, and versatility. Polyethylene plastic is used in packaging, construction, and medical products. The material is used to make plastic bags, containers, and bottles for packaging. Polyethylene is applied in pipes, insulation, and geomembranes because of its strength and resistance to moisture and chemicals in construction. Specific grades of polyethylene in the medical field, such as medical-grade HDPE and ultra-high-molecular-weight polyethylene (UHMWPE), are used for products (surgical drapes and medical tubing) due to their durability, chemical resistance, and biocompatibility.
What is Polyethylene?
Polyethylene is classified into low-density polyethylene (LDPE) and high-density polyethylene (HDPE) based on molecular structure. LDPE has a branched structure that makes it flexible and less dense, suitable for plastic bags and squeeze bottles. HDPE has a more linear structure that provides higher strength, rigidity, and chemical resistance, making it ideal for items (milk jugs, water pipes, and plastic toys). The differences in molecular structure give LDPE and HDPE distinct physical properties and applications.
How is Polyethylene Made?
Polyethylene is made by the polymerization of ethylene gas (C₂H₄) using heat, pressure, and catalysts to link the molecules into long polymer chains. The polyethylene manufacturing process determines whether the product is low-density polyethylene (LDPE) or high-density polyethylene (HDPE). LDPE is produced by free-radical polymerization at very high pressure and temperature, creating a branched structure that prevents tight packing, resulting in a flexible and less dense material. HDPE is made using catalysts such as Ziegler–Natta or Phillips systems under lower pressure, forming linear chains that pack closely, giving higher strength and density. These structural differences define their properties and applications.
What is the Full Form of PE?
The full form of PE is Polyethylene. Polyethylene is a thermoplastic polymer made from polymerized ethylene monomers. The material is one of the most used plastics due to its versatility, low cost, and ease of production. Polyethylene is found in packaging materials (plastic bags, bottles, and containers). Polyethylene is used in the manufacturing of pipes, insulation, and construction materials. The polymer’s durability and resistance to chemicals make it a preferred choice for various applications in industries (packaging, construction, and medical devices). The polyethylene full form is PE, and the PE material is utilized across many sectors, including packaging (plastic bags, bottles), construction (pipes, geomembranes), and healthcare (medical tubing, surgical drapes).
What are the Main Challenges in Manufacturing Polyethylene?
The main challenges in manufacturing Polyethylene are listed below.
- High energy consumption: Polyethylene production requires significant energy input due to the high temperatures and pressures involved in the polymerization process, which contributes to increased operational costs and environmental impact. Polyethylene manufacturing challenges include addressing energy efficiency and minimizing consumption during the production phase.
- Environmental impact: The production of polyethylene generates waste and emissions, including greenhouse gases. The disposal of polyethylene products, which are non-biodegradable, contributes to long-term environmental pollution. Polyethylene production issues revolve around reducing waste and mitigating the negative environmental effects of polyethylene products.
- Material quality control: Maintaining consistent quality during polyethylene production is challenging. Variations in the polymerization process lead to differences in molecular structure, which affect the final material's properties (strength, flexibility, and durability). The control of the variations is essential to ensuring uniform product quality.
How does Polyethylene Affect the Quality of Products?
Polyethylene affects the quality of products by increasing their durability, polyethylene product quality, impact resistance, and low-friction properties. Polyethylene's high strength-to-weight ratio makes it resistant to physical wear, providing long-lasting performance in various applications (packaging materials, automotive parts, and construction components). The material's polyethylene impact resistance helps products maintain their integrity even under rough conditions, preventing cracks and damage. Polyethylene's low-friction properties reduce wear and tear, increasing the lifespan of components that experience frequent motion. Polyethylene contributes to the quality of products by ensuring protection during transportation and handling in packaging. The material's durability and flexibility make it ideal for creating sturdy yet lightweight packaging materials. Polyethylene improves the performance and longevity of components (fuel tanks and bumpers) by offering resistance to impacts and harsh environmental conditions in automotive parts. Polyethylene is used in pipes, insulation, and geomembranes due to its strength and resistance to corrosion in construction materials, ensuring the materials remain functional over time, even in challenging environments. The characteristics make polyethylene a critical material for applications where durability, performance, and longevity are essential.
What are the Advantages of Polyethylene?
The advantages of Polyethylene are listed below.
- Cost-effective: Polyethylene is an affordable material to produce, making it a popular choice for industries seeking budget-friendly solutions. Polyethylene's low production costs help keep the prices of products like plastic bags, bottles, and packaging materials affordable. The uses of polyethylene in packaging, containers, and household products benefit from the cost savings.
- Versatile: Polyethylene is adapted for a wide range of applications (packaging, construction, and the medical industries). Polyethylene's ability to be molded into different shapes and forms, and its availability in various grades, make it suitable for a variety of uses.
- Lightweight: Polyethylene is lightweight and strong, which makes it ideal for products requiring strength without adding significant weight. The material's lightness is beneficial in packaging and transportation, reducing overall shipping costs. Polyethylene advantages in transportation and logistics are substantial in reducing fuel consumption due to its reduced weight.
- Durable: Polyethylene offers excellent resistance to chemicals, moisture, and environmental conditions. The durability makes it a preferred material in harsh environments (for pipes in construction and outdoor applications).
- Flexible: Low-density polyethylene (LDPE) is highly adaptable and resistant to cracking, making it ideal for packaging materials (plastic bags) and for applications where flexibility is essential.
What are the Disadvantages of Polyethylene?
The disadvantages of Polyethylene are listed below.
- Environmental concerns: Polyethylene is non-biodegradable, meaning it persists in the environment for hundreds of years. Improper disposal contributes to pollution in oceans and landfills, threatening wildlife and ecosystems. The polyethylene environmental impact is significant due to the large amounts of plastic waste that accumulate and the difficulty in recycling the material.
- Low temperature sensitivity: Polyethylene becomes brittle at low temperatures, which causes it to crack or break under stress. The result limits its use in environments with extreme cold temperatures.
- UV degradation: Exposure to ultraviolet (UV) light degrades polyethylene over time, causing it to become brittle, discolored, and weakened. The polyethylene disadvantage is its reduced durability in outdoor applications without proper UV stabilizers or coatings.
Polyethylene is a family of simple, carbon–hydrogen polymers whose tunable molecular architecture enables grades ranging from soft, highly flexible films to rigid, pressure-capable pipes and structural parts. Its combination of low cost, chemical resistance, moisture impermeability, and low friction underpins dominant use in packaging, construction, and piping, while ultra-high-molecular-weight grades extend its role into demanding tribological and biomedical applications such as joint implants. Ease of processing makes polyethylene the default material for many high-volume products, though this same ubiquity amplifies end-of-life challenges due to its persistence, microplastic formation, and imperfect recycling systems. In performance terms, polyethylene outperforms many commodity plastics in toughness, low-temperature behavior, and moisture resistance, but trades off UV resistance, stiffness, and gas-barrier properties, which shapes where it can be reliably specified.
What are the Properties of Polyethylene?
The properties of polyethylene are listed below.
- Chemical resistance: Polyethylene is resistant to a wide range of chemicals, including acids, bases, and solvents. Polyethylene makes it ideal for applications in the chemical and pharmaceutical industries, where it must withstand exposure to various substances without degrading.
- Flexibility: Polyethylene, low-density polyethylene (LDPE), is highly flexible, allowing it to be easily molded into different shapes and forms. The property is beneficial in packaging, where flexibility is essential for creating products like plastic bags and stretch films.
- Insulating properties: Polyethylene has excellent electrical insulation, making it suitable for electrical wiring and cables. Polyethylene's ability to prevent the flow of electricity helps ensure safety and reliability in electrical applications.
- Durability: Polyethylene has high resistance to impact, abrasion, and environmental conditions, making it durable in harsh conditions. Polyethylene durability is essential in construction, where polyethylene is used in pipes, geomembranes, and insulation materials.
- Low friction: Polyethylene has a low coefficient of friction, making it ideal for applications involving sliding or moving parts (such as conveyor systems and packaging machinery). The polyethylene properties of low friction contribute to the efficient operation of many industrial processes.
Planning to Use Polyethylene For Your Parts?
What is the Density of Polyethylene?
The density of polyethylene varies by type, with low-density polyethylene (LDPE) ranging from 0.91 to 0.93 g/cm³ and high-density polyethylene (HDPE) ranging from 0.94 to 0.97 g/cm³. The difference in density between LDPE and HDPE is due to their molecular structures. LDPE has a more branched structure, which results in lower packing efficiency and a lower density, while HDPE has a more linear and tightly packed structure, contributing to its higher density. The polyethylene density affects its flexibility, strength, and suitability for various applications (packaging materials, plastic pipes, and containers), where different densities are required for flexibility or durability. LDPE is more flexible and less rigid due to its lower density, making it ideal for products that require stretchability (plastic bags and film wraps). HDPE is stronger and more rigid due to its high density, making it suitable for products that require strength and durability (containers, pipes, and rigid plastics). The differences in density between LDPE and HDPE influence the material's performance and determine their suitability for specific uses in industries ranging from packaging to construction.
What is the Melting Point of Polyethylene?
The melting point of polyethylene varies by type, with low-density polyethylene (LDPE) ranging from 105°C to 115°C and high-density polyethylene (HDPE) between 120°C and 130°C. The difference in melting points is due to the molecular structure of each type. LDPE has a more branched structure, which prevents the molecules from packing closely together, resulting in a lower melting point. HDPE has a more linear and tightly packed structure, leading to a higher melting point and greater strength. The polyethylene melting point impacts its processing and molding. A lower melting point, like that of LDPE, allows for easier processing at lower temperatures, making it suitable for applications that require flexibility and lightweight properties. HDPE, with its higher melting point, requires higher processing temperatures but offers enhanced strength and rigidity, making it ideal for applications requiring durability, such as piping and containers. The differences in melting points influence the choice of polyethylene type for specific industrial applications.
What is the Chemical Formula of Polyethylene?
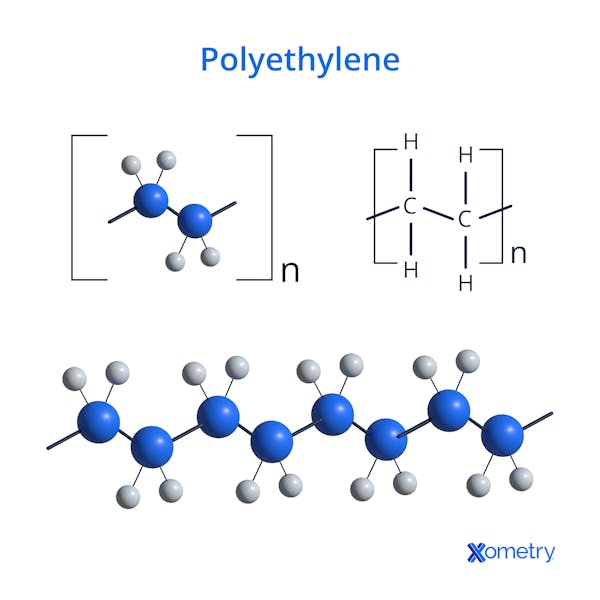
The chemical formula of polyethylene is (C₂H₄)n. Polyethylene is formed by polymerizing ethylene monomers (C₂H₄), where each ethylene unit consists of two carbon atoms bonded to four hydrogen atoms. The polymerization process links the ethylene molecules into long chains, with the number of repeating units (n) determining the polymer's molecular weight. The process results in a long, flexible polymer chain with consistent chemical properties, making the polyethylene chemical formula an essential factor in its wide range of industrial applications (packaging and construction).
What is the Structure of Polyethylene?
The structure of polyethylene is a simple, linear arrangement of carbon atoms bonded to hydrogen atoms, formed by the polymerization of ethylene monomers. Each ethylene unit consists of two carbon atoms and four hydrogen atoms, and the units repeat to form long, unbranched chains. The polyethylene structure differs between low-density polyethylene (LDPE) and high-density polyethylene (HDPE). LDPE has a branched structure, where the polymer chains are irregular and less tightly packed. The branching results in a more flexible, less dense material. HDPE has a more rigid, crystalline structure due to its linear arrangement, allowing the chains to pack more tightly, creating a stronger and denser material. The structural differences contribute to the distinct physical properties of LDPE and HDPE, making them suitable for various applications (plastic bags and packaging for LDPE, and pipes and containers for HDPE).
What is Polyethylene Used for?
Polyethylene is used for a variety of applications (packaging materials, piping systems, and medical devices). Polyethylene is used for plastic bags, bottles, and containers due to its flexibility, durability, and low cost. The properties make polyethylene an ideal packaging material, as it provides a lightweight and protective barrier for products. Polyethylene uses are extensive in piping systems (water and gas pipes). Polyethylene's chemical resistance, flexibility, and ability to withstand harsh environmental conditions make it perfect for use in infrastructure projects where durability and corrosion resistance are essential. Polyethylene applications include devices (syringes, medical tubing, and surgical drapes) in medical applications. The material's biocompatibility, resistance to moisture, and ease of sterilization make it suitable for ensuring safety and reliability in medical settings. The unique combination of properties makes polyethylene ideal for the applications, offering the necessary performance and cost-effectiveness required across diverse industries.
What is Polyethylene Used for in Medical Applications?
Polyethylene is used in medical applications for implants, packaging, and prosthetics. Polyethylene is utilized due to its durability and ability to withstand bodily conditions in implants. Packaging for medical devices is made from polyethylene, which provides a sterile and protective barrier. Polyethylene is used in joint replacements for prosthetics in hip and knee prostheses, as it withstands wear and tear over time while maintaining structural integrity. Polyethylene’s biocompatibility makes it suitable for medical applications, as it does not cause adverse reactions when in contact with the human body. The material's resistance to bacteria ensures that polyethylene used in medical products remains hygienic and free from contamination. The material’s durability helps in medical devices, as it endures long-term use without significant degradation, ensuring reliability and safety in critical applications.
Can Polyethylene be Used in Medical Implants?
Yes, polyethylene can be used in medical implants. Polyethylene is used in orthopedic implants for load-bearing joints (hip and knee replacements). The durability, wear resistance, and biocompatibility of polyethylene make it an excellent choice for such applications. Polyethylene provides a long-lasting solution in joint replacements, as it withstands the mechanical stresses and friction involved in movement without degrading. The material’s ability to resist bacterial contamination and its smooth surface further contribute to its effectiveness in medical implants, ensuring functionality and safety for the patient.
What are the Applications of Polyethylene in Construction?
The applications of polyethylene in construction are listed below.
- Water pipes: Polyethylene is used for water distribution pipes due to its resistance to corrosion, flexibility, and ease of installation. The polyethylene properties help ensure longevity and reliability in water systems, even in harsh environments.
- Geosynthetics: Polyethylene is used in soil stabilization, erosion control, and drainage systems because of its strength, durability, and moisture resistance. The polyethylene properties of flexibility and resistance to environmental factors make it ideal for reinforcing soil and preventing erosion in construction projects.
- Damp proofing: Polyethylene serves as a damp-proof membrane to prevent moisture from penetrating foundations and ensures structural longevity. The polyethylene properties of being impermeable to water and resistant to mold growth make it perfect for keeping structures dry and stable.
- Insulation: Polyethylene is used in foam insulation boards and under-slab vapor barriers, providing low thermal conductivity and temperature control. The polyethylene properties of being lightweight and having excellent insulating abilities make it ideal for maintaining energy efficiency in buildings.
- Underlayments for flooring: Polyethylene sheets act as moisture barriers, protecting flooring from water damage and mold growth. The polyethylene properties of moisture resistance and durability make it an effective solution for safeguarding floors, especially in areas exposed to high humidity.
What Role does Polyethylene Play in Automotive Safety Features?
The role of polyethylene in automotive safety features is significant in bumpers. Polyethylene is used in bumpers due to its strength, flexibility, and ability to absorb energy during a collision, reducing impact forces in low-speed crashes and protecting the vehicle and its occupants. Polyethylene's lightweight and durable nature allows for fast and reliable inflation during a collision in airbags. The strength, flexibility, and energy absorption properties of polyethylene help minimize injury by cushioning impact forces. Polyethylene's ability to deform without breaking ensures that safety features (bumpers and airbags) continue to perform effectively under stress. The attributes make polyethylene an ideal material for improving vehicle safety in the event of a collision.
What are the Benefits of Polyethylene in Food Packaging?
The benefits of polyethylene in food packaging are listed below.
- Moisture resistance: Polyethylene effectively prevents moisture from entering or exiting packaged food, helping to maintain the food's freshness and extend its shelf life. The property is beneficial for items (bread, fruits, and vegetables).
- Chemical inertness: Polyethylene is chemically inert, meaning it does not react with most foods. The process ensures that the food remains uncontaminated and retains its original taste and quality during storage.
- Lightweight: Polyethylene is lightweight, which reduces shipping costs and makes it easier to handle. Polyethylene's lightness makes it ideal for creating flexible packaging (bags, wraps, and pouches).
- Durability: Polyethylene is durable and resistant to tears and punctures, making it an ideal material for packaging items that need protection during transportation and handling (frozen foods, snacks, and liquids).
- Flexibility: Polyethylene is highly flexible, allowing it to be molded into various shapes and sizes, making it suitable for different types of food packaging (vacuum-sealed bags and shrink wraps).
- Cost-effectiveness: Polyethylene is inexpensive to produce, keeping food packaging costs low. The cost-effectiveness makes it a popular choice for manufacturers in the food industry for high-volume packaging.
- Recyclability: Polyethylene is recyclable, helping to reduce environmental impact. Polyethylene products are recycled and repurposed into new packaging materials, contributing to sustainability efforts in the food industry.
UHMWPE is known for its superior strength and wear resistance, making it suitable for demanding applications, whereas LLDPE is valued for its flexibility, ease of processing, and use in everyday applications like packaging and films.Kurt PokopacSolutions Engineer
What is the Cost of Polyethylene Compared to Other Plastics?
The cost of polyethylene to other plastics is lower, ranging from US[ $1.50 to US $3.00] per kilogram for virgin resin. The affordability is due to polyethylene's simple manufacturing process and the abundant availability of ethylene, the raw material used in its production. Polyethylene requires less energy to produce than other plastics, which helps keep its production costs down. Polypropylene and polystyrene require more complex processes and higher raw material costs, making them more expensive to produce. The cost-effectiveness of polyethylene contributes to its widespread use in various industries (packaging, construction, automotive, and consumer goods), where its affordability and versatility provide significant cost savings. Polyethylene's low production cost makes it ideal for high-volume applications, such as packaging, where cost savings are crucial. The affordability has led to polyethylene’s dominance in sectors (food packaging, automotive parts, and construction materials), ensuring that it remains a popular and economically viable material. The combination of low cost and versatile properties makes polyethylene a preferred material across a wide range of applications.
How does Polyethylene Compare to Other Plastics in Terms of Durability?
Polyethylene is durable compared to other plastics in terms of durability by offering superior resistance to moisture, chemicals, and impacts. High-density polyethylene (HDPE) is strong, making it suitable for applications requiring high strength (water pipes, containers, and outdoor products). Low-density polyethylene (LDPE), while more flexible, is less durable and prone to wear and tear, making it ideal for lighter-duty applications (plastic bags and wraps). Polyethylene is more resistant to environmental factors than other plastics (polypropylene and polystyrene). Polyethylene withstands chemicals and moisture better than alternatives (polypropylene and polystyrene), making it ideal for applications (water pipes, chemical containers, and agricultural films). Polyethylene's durability, flexibility, and resistance to harsh conditions make it a versatile and long-lasting material in numerous industries.
Is Polyethylene (PE) More UV-Resistant than Polycarbonate (PC)?
No, polyethylene (PE) is not more UV-resistant than polycarbonate (PC). Polycarbonate offers better protection against ultraviolet (UV) degradation due to its inherent ability to resist UV light, which helps prevent the material from becoming brittle or discolored over time. PE is more susceptible to UV degradation, which leads to a breakdown of the polymer chains when exposed to prolonged sunlight. UV stabilizers are added during the manufacturing process to improve the UV resistance of PE. The stabilizers help reduce the material's sensitivity to UV light, but do not provide the same level of protection as polycarbonate. The Polycarbonate inherently possesses stronger UV resistance, which makes it more suitable for outdoor applications where prolonged exposure to sunlight is a concern. PE's lower UV resistance limits its use in such applications unless UV stabilizers are applied.
Is Polyethylene (PE) Easier to Mold than Acrylonitrile Butadiene Styrene (ABS)?
Yes, polyethylene (PE) is easier to mold than acrylonitrile butadiene styrene (ABS). Low-density polyethylene (LDPE) is easy to mold due to its flexibility, lower melting point, and more amorphous molecular structure, which allows it to flow readily during the molding process. The property reduces the energy required and simplifies the equipment needed for shaping, making LDPE ideal for high-volume, cost-effective molding applications. High-density polyethylene (HDPE) is slightly more challenging to mold than LDPE because of its higher melting point and more crystalline structure, but it is easier to mold than ABS. ABS has a higher melting point and a rigid molecular structure, demanding more precise temperature control and processing conditions. LDPE is ideal for simple molding due to its lower melting point and flexible structure, while Acrylonitrile Butadiene Styrene (ABS) is preferred for applications requiring high strength and dimensional stability.
What Properties of Polyethylene Make It a Good Electrical Insulator?
The properties of polyethylene make it a good electrical insulator, including its non-polar molecular structure and the absence of free charge carriers, which prevent the flow of electricity. PE polyethylene properties, such as high-density polyethylene (HDPE), with higher crystallinity and density than low-density polyethylene (LDPE), provide slightly higher dielectric strength and better mechanical durability. Polyethylene properties (LDPE) are more flexible but have lower dielectric strength. The High-Density Polyethylene (HDPE) is ideal for electrical insulation in cables, wires, and other applications requiring durability and performance.
How is Polyethylene Recycled?
Polyethylene is recycled by collecting, sorting, and cleaning the material to remove contaminants before it is melted and reformed into pellets or granules for new products. The process reduces polyethylene waste in landfills and conserves resources by reusing the material. Low-density polyethylene (LDPE) and high-density polyethylene (HDPE) differ in recycling behavior due to their molecular structures. LDPE has a lower melting point and flexible chains, making it easier to process in some cases, but thin films and bags do not tangle in machinery. HDPE has a more rigid and crystalline structure, requiring higher temperatures for melting but allowing smoother industrial processing. Contamination from food residue or non-polyethylene materials lowers the quality of recycled products and complicates sorting. Polyethylene recycling is essential for polyethylene sustainability, reducing environmental impact and conserving raw materials.
What Are the Environmental Impacts of Polyethylene Production and Disposal?
The environmental impacts of polyethylene production and disposal are listed below.
- Energy consumption: The production of polyethylene requires significant energy input during the polymerization process. The process contributes to greenhouse gas emissions and higher operational costs, increasing the environmental footprint.
- Pollution: Polyethylene production generates waste and emissions (volatile organic compounds and greenhouse gases). Inadequate waste management during production results in harmful environmental effects.
- Non-biodegradability: Polyethylene is non-biodegradable and persists in landfills for hundreds of years. The accumulation in the environment in oceans contributes to long-term pollution and harm to wildlife.
- Microplastic contamination: Polyethylene breaks down into microplastics that contaminate soil, water, and marine ecosystems. The microplastics are harmful to aquatic life and enter the food chain.
- Recycling challenges: Polyethylene is recyclable, but contamination and inefficient sorting processes limit its effectiveness. A large percentage of polyethylene products are not recycled, contributing to environmental waste.
Is Polyethylene (PE) More Environmentally Friendly than Polylactic Acid (PLA)?
No, polyethylene (PE) is not more environmentally friendly than polylactic acid (PLA), but the comparison depends on context. PLA is made from renewable resources, such as corn starch or sugarcane, and is biodegradable under industrial composting conditions. Food packaging and single-use items that are compostable make it a better choice when composting is available. Polyethylene is a petroleum-based plastic that does not biodegrade under normal environmental conditions and persists for decades in landfills and oceans. HDPE and LDPE are recyclable, but recycling rates vary based on local facilities, contamination, and sorting practices. PLA is not a perfect solution despite its benefits. The material's biodegradability is limited outside industrial composting, and its production requires land, water, and energy, which can have environmental impacts. PLA interferes with conventional plastic recycling streams if not properly separated. The Polylactic Acid (PLA) has advantages in renewable sourcing and industrial compostability, while polyethylene’s durability and established recycling infrastructure provide different environmental trade-offs. The most environmentally friendly choice depends on the specific use case and available waste management infrastructure.
Is Polyethylene (PE) More Easily Recycled than Acetal (POM)?
Yes, polyethylene (PE) is more easily recycled than Acetal (POM). Polyethylene is recycled through mechanical processes, where it is shredded, melted, and reformed into new products. The recycling process is well-established, supported by a broad infrastructure, and common in many regions. PE is remelted and reshaped multiple times, although repeated recycling reduces its mechanical properties. Recycling Acetal (POM) is more challenging due to its higher melting point and crystalline structure, which make it more stable and durable but harder to process for recycling. Specialized equipment is required, and recycling infrastructure for POM is limited, resulting in lower recycling rates compared to polyethylene. POM is chemically recycled, but it is not widely implemented. Polyethylene’s broader recycling infrastructure and easier processing make it easier to recycle than Acetal (POM).
Does Polyethylene (PE) Offer Better Moisture Resistance than High Impact Polystyrene (HIPS)?
Yes, polyethylene (PE) offers better moisture resistance than high-impact polystyrene (HIPS). Polyethylene is hydrophobic, meaning it repels water, which makes it well-suited for applications (packaging and piping) where moisture resistance is essential. HIPS absorbs more water than PE despite being durable and impact-resistant. HIPS contains a rubbery butadiene phase, which absorbs small amounts of moisture over time. The molecular structure of polyethylene, with long non-polar polymer chains, allows it to resist moisture more effectively than the more polar components of HIPS. Polyethylene is a better choice for applications where moisture resistance is critical, although High Impact Polystyrene (HIPS) provides moderate resistance and remains suitable for many other uses.
How Does Polyethylene (PE) Perform in Moisture-Exposed Environments Compared to Other Plastics?
Polyethylene (PE) performs well in moisture-exposed environments compared to other plastics by offering superior resistance to water absorption. PE has a hydrophobic nature, which makes it ideal for uses such as pipes, packaging, and containers. The material does not degrade or lose structural integrity under normal exposure to water, making it one of the most reliable materials in moisture-prone environments. Polypropylene (PP) and polyvinyl chloride (PVC) perform well in wet conditions, while polystyrene (PS) is moderately resistant. PS is brittle and less suitable for structural applications in wet environments, whereas PVC is highly water-resistant but chosen for different mechanical properties. PE’s combination of hydrophobicity, durability, and versatility makes it a preferred choice for industries (construction, food packaging, automotive, and healthcare).
Does Polyethylene (PE) Exhibit Better Low-Temperature Performance than Polyethylene Terephthalate (PET)?
Yes, polyethylene (PE) exhibits better low-temperature performance than polyethylene terephthalate (PET). PE remains flexible at much lower temperatures, while PET tends to become brittle in cold conditions due to its more rigid and crystalline structure. The molecular structure of PE allows it to retain flexibility even at sub-zero temperatures, making it suitable for applications that require resilience in cold environments (frozen food packaging, outdoor films, and flexible or insulated pipes). The Polyethylene Terephthalate (PET) is more rigid and is used in applications (bottles, food containers, and textile fibers) where low-temperature flexibility is less critical. The distinct low-temperature performance of the materials helps determine their suitability for different uses across industries.
Is Polyethylene (PE) More Resistant to Abrasion than Nylon (PA)?
No, polyethylene (PE) is not more resistant to abrasion than nylon (PA). Nylon offers superior abrasion resistance due to its higher tensile strength and more rigid molecular structure, making it ideal for applications (textiles, ropes, conveyor belts, and automotive parts). Polyethylene is durable and impact-resistant but wears more quickly under repeated friction. Polybutylene terephthalate (PBT) provides good gas barrier properties, making it suitable for food packaging applications (bottles, trays, and containers), where protection from oxygen and moisture helps extend shelf life. The durability and barrier properties of PE and PBT make them suitable for specific applications, whereas Nylon (PA) is preferred in industries requiring abrasion resistance.
Does Polyethylene (PE) Have Better Barrier Properties Against Gases than Polybutylene Terephthalate (PBT)?
No, polyethylene (PE) does not have better gas barrier properties than polybutylene terephthalate (PBT). PBT provides superior resistance to gases like oxygen, making it the preferred choice for applications that require protection, such as food packaging, where it helps preserve freshness and extend shelf life. PE is more permeable and less effective in environments where gas resistance is critical. The Polybutylene Terephthalate (PBT) has a higher melting point and greater thermal stability than PE, making it suitable for high-temperature applications(automotive and electrical components). PE softens above its melting point and is better suited for low- to moderate-temperature uses, including flexible packaging and low-pressure pipes.
How Xometry Can Help
As you can see, polyethylene is a versatile material with multiple useful variants. At Xometry we offer instant quoting of machined or sheet-cut polyethylene parts, specifically UHMWPE and HDPE. If you’re looking for high-volume production capabilities, we’ve got you covered with our injection molding services. Choose from polyethylene or from dozens of other materials through the Xometry Instant Quoting Engine® today!
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.


