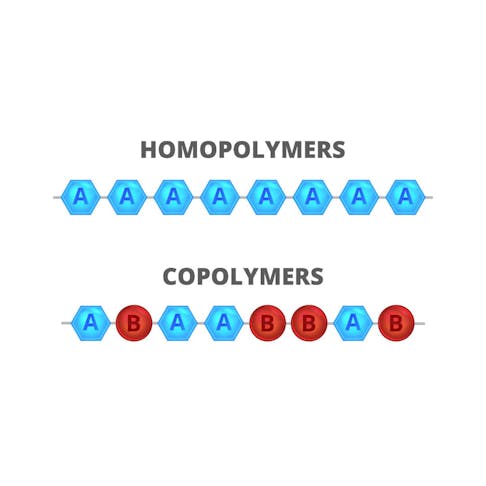
Copolymers and homopolymers are distinct groups of polymers defined by the makeup and arrangement of their chemical structures. Homopolymers are distinguished by the fact that they are formed of one type of repeated monomer. Conversely, copolymers are made of at least two variants of monomers in any form of repeating pattern. While copolymers and homopolymers have some varying mechanical properties they mostly share the same applications, including: textiles, piping, car trim, films, and battery cases.
This article will discuss the differences between copolymer vs homopolymers, in terms of types, applications, advantages, and disadvantages.
What Is Homopolymer?
A homopolymer is a polymer in which all monomer units are identical. Polymers are a material which has a chemical structure consisting of a large chain of monomers. Some examples of homopolymers are polyvinyl chloride, which is constructed of multiple vinyl chloride units, and polypropylene, which is made of repeated propylene units.
What Is the Purpose of Homopolymer?
The purpose of homopolymers is to produce a polymer that has increased mechanical properties. These enhanced mechanical properties include: tensile strength, stiffness, impact resistance, and short-term creep resistance.
Where Are Homopolymers Used?
Homopolymers are used in a range of polymer manufacturing techniques including: injection molding, blow molding, thermoforming, and film, fiber, or sheet extrusion. These processes produce homopolymers for several industries including: automotive, packing, medical, consumer goods, and food containers. Within these industries, the homopolymers' corrosion resistance makes them ideal for containers, tanks, and pipes. Its dimensional stability allows it to be used for gears and bearings. Homopolymer’s moisture resistance makes it a suitable choice for making clothes, carpets, fibers, and films.
What Are the Types of Homopolymer?
Homopolymers are classified by the fact that they are produced from a single variant of monomer, but each type of homopolymer may have a different monomer. The types of homopolymers include: polyvinyl chloride, polyethylene, polypropylene, polycarbonate, polyester, nylon 6, nylon 11, polytetrafluoroethylene, and styrene.
What Are the Advantages of Homopolymer?
The advantages of a homopolymer mainly lie in its mechanical properties, which are highlighted below:
- High mechanical strength.
- High stiffness.
- High hardness.
- High initial creep resistance.
- Increased wear resistance.
- Low rate of thermal expansion.
What Are the Disadvantages of Homopolymer?
The disadvantages of homopolymers are:
- Poor ultraviolet resistance.
- Poor acid or alkali resistance.
- Poor thermo-oxidative stability.
- Poor fire resistance.
What Is Copolymer?
A copolymer is a polymer made up of more than one type of monomer unit. Copolymers are produced by polymerizing two or more types of monomer together in a process referred to as copolymerization. Copolymers produced in this way are sometimes also referred to as biopolymers.
What Is the Purpose of Copolymer?
The purpose of creating a copolymer is to manufacture a polymer with more desirable properties. Copolymers typically experience a lower crystallinity, higher glass transition temperature, and improved solubility. These characteristics are achieved through a process referred to as rubber toughening.
Where Are Copolymers Used?
Copolymers can be found in many applications, including in: drug delivery, cosmetics, suncream, liquid membranes, emulsifiers, hoses, textiles, bearings gears, and self-polishing paint. Copolymers are commonly hygroscopic in nature, which accounts for some of the less-expected applications listed.
What Are the Types of Copolymer?
All copolymers may consist of two or more types of monomers. The different monomers can repeat singularly, in blocks, or in some other statistically repeatable way. Types of copolymers include: polyvinyl acetate, ethylene-vinyl acetate, high-impact polystyrene, acrylonitrile butadiene styrene, styrene-butadiene rubber, styrene-acrylonitrile rubber, styrene butadiene styrene, and polyethylene oxide.
What Are the Advantages of Copolymer?
The advantages of using copolymers include:
- High shear resistance.
- High operating temperature.
- High corrosion resistance.
- High impact resistance.
- High dimensional stability.
What Are the Disadvantages of Copolymer?
There are some disadvantages to choosing copolymers over homopolymers, which are listed below:
- Poor wetting characteristics.
- Expensive.
- Comparatively poor mechanical properties.
Which Is Better, Homopolymer or Copolymer?
It depends. Neither homopolymer nor copolymer is better than the other. It will depend on the application as to which is more desirable. Homopolymers are better suited to high-strength and high-hardness applications, whereas copolymers are suited for corrosion resistance and high operating temperatures. Although a raw homopolymer has better mechanical properties, copolymers have better mechanical properties when reinforced with glass over glass-reinforced homopolymer. This is because copolymers are better suited to adhering to fibers.
How Do Copolymers and Homopolymers Differ in Structure?
Copolymers and homopolymers differ in their chemical structure. Homopolymers consist of one variant of monomer which is repeated multiple times in a chain. Copolymers differ from homopolymers in structure because they use two or more variants of monomer to produce a chain.
Additionally, copolymers and homopolymers differ in the way their monomer chains are connected. Copolymers are differentiated into linear copolymers and branched copolymers.
These two categories are further divided into alternating, statistical, and block for linear copolymers, and grafted and star for branched copolymers.
An alternating linear copolymer alternates consecutively as in Figure 2 (A-B-A-B-A-B). A block linear copolymer will alternate in blocks of repeating monomers (A-A-A-A-B-B-B-B). Lastly, a statistical linear copolymer comprises two or more repeating monomers that repeat in a statistically predictable pattern.
What Are the Properties of Copolymer and Homopolymer?
Table 1 compares various properties of a generic copolymer and homopolymer:
| Property | Copolymer (Generic) | Homopolymer (Generic) |
|---|---|---|
Property Density | Copolymer (Generic) 0.9 g/cm³ | Homopolymer (Generic) 0.9 g/cm³ |
Property Tensile Strength | Copolymer (Generic) 60 MPa | Homopolymer (Generic) 69 MPa |
Property Tensile modulus | Copolymer (Generic) 950 N/mm² | Homopolymer (Generic) 1,600 N/mm² |
Property Tensile stress | Copolymer (Generic) 25 N/mm² | Homopolymer (Generic) 37 N/mm² |
Property Melting point | Copolymer (Generic) 160–165 °C | Homopolymer (Generic) 135–159 °C |
The table demonstrates how both polymers have the same density but the homopolymer has a higher tensile strength, strain, and stress.
What Is the Strength of Homopolymer Compared to Copolymer?
The tensile strength of a homopolymer is, on average, 15% higher than that of a copolymer. This reduction in strength for the copolymer comes from the reduced crystallinity. The better fatigue resistance and creep resistance of a homopolymer are also attributable to the higher level of crystallinity.
What Are the Applications of Copolymer and Homopolymer?
While copolymers and homopolymers do have some different properties, they do also share most applications. Some shared applications include: packaging, textiles, tubing, films, furniture, battery cases, car trim, and liquid tanks.
Is Polythene a Copolymer?
No, polythene is not a copolymer. Polythene is a homopolymer as it is produced through the polymerization of a single monomer called ethylene. As the definition of a homopolymer is a polymer made of a single repeating monomer unit, then polythene is a homopolymer.
To learn more, see our article on Polyethylene Composition.
Is Nylon 6 a Copolymer?
No, nylon 6 is not a copolymer. The clue is in the name “nylon 6” in which the 6 represents the single repeating monomer which has 6 carbon atoms. Nylon 6 is made through the polymerization of a monomer named caprolactam. Nylon 6 is not to be confused with nylon 6,6 which is made of two repeating monomers hexamethylenediamine and adipic acid; this makes it a copolymer. Two other nylons are also copolymers; they are nylon 6,12 and nylon 4,6.
Is PVC a Homopolymer?
Yes, PVC is a homopolymer. The single repeating monomer unit of polyvinyl chloride is vinyl chloride, the “Poly” in PVC means multiple. Hence PVC is made up of multiple repeating units of vinyl chloride.
Summary
This article presented copolymers and homopolymers, explained each of them, and discussed their key differences. To learn more about copolymers and homopolymers, contact a Xometry representative.
Xometry provides a wide range of manufacturing capabilities and other value-added services for all of your prototyping and production needs. Visit our website to learn more or to request a free, no-obligation quote.
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.