Ductility is a material property that describes the deformation under tensile stress by inelastic stretching, after the yield point and up to fracture. Ductility is measured either by the percentage elongation or percent reduction in cross-sectional area that takes place before a fracture occurs, under tensile stress. Materials that are highly ductile, such as copper, gold, and aluminum, can undergo significant plastic deformation before a fracture happens. Ductility is a key property in the working of metals, and in concert with malleability, it is the basis for much of the shaping and forming of metals that allows complex parts to be made. This article will discuss six of the most ductile metals, how ductility is measured, what affects it, and where ductile metals are used.
1. Gold
Gold of high purity was long considered the most ductile metal. This property allows it to be stretched into extremely thin wires or elongated into thin sheets/foils without undergoing brittle fracture. Gold's high ductility is attributed to its face-centered cubic crystalline structure. This facilitates dislocations and reformation of bonds, as atoms in the metal slide past one another breaking and reforming the (electrostatic or weak atomic) bonds between them.
Overall, the ductility of gold is a valuable property that makes the metal useful in a wide range of applications, from electrical wiring to medical implants.
2. Platinum
Platinum is demonstrably the most ductile metal. A single gram of platinum can be drawn into a wire many kilometers long. This property makes platinum very useful in a variety of applications, including electrical contacts, jewelry, and catalytic converters in cars. Platinum's high ductility is also due to its face-centered cubic atomic lattice structure, which allows the same behavior as gold.
3. Copper
Copper is another metal with a face-centered cubic atomic structure that is highly ductile. As it is also highly conductive, plentiful, and very easily refined, it is ideal for manufacturing electrical wires. For more information, see our guide on Copper.
4. Nickel
Nickel is a highly ductile material that can be drawn into wires and sheets without fracturing. This is directly related to the face-centered cubic atomic structure that allows bonds to break and reform easily. Nickel is used in high-temperature applications because of its retention of strength at elevated temperatures.
5. Niobium
Niobium is a soft, gray metal that is highly ductile and is unusual in the ductility list in that it has a body-centered cubic crystal structure. It is not an important engineering material on its own but an alloying agent. It improves the strength of steel and stainless steel by lending some of its ductility to the resultant properties.
6. Tantalum
Tantalum is a highly ductile material, well suited to making complex parts and profiles. Its corrosion resistance makes it an attractive option in aggressive environments and its workability is attractive when forming complex shapes. It is widely used in reactors, aggressive chemical handling, and heat exchangers.
What Does Ductile Metal Mean?
If metal is ductile, it can undergo significant deformation before a fracture happens. The technically correct definition of the ductility of a material is either the percentage elongation or percentage cross-sectional area reduction a material can withstand while undergoing pure tensile loading.
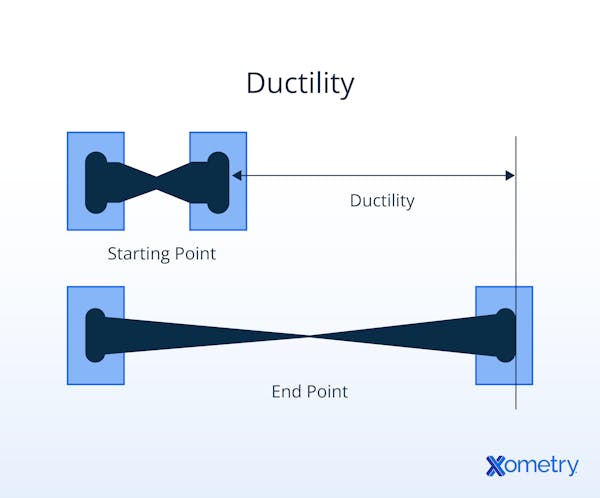
What Makes a Metal Ductile?
Metals that have a simple face-centered cubic crystalline structure and tend to form large regions of crystallization are generally more ductile. Stacked layers of atoms can slide over each other along slip planes that are joined by the weak atomic (electrostatic) force, where bonds reform as a new lattice position is reached. Where crystal growth regions are smaller, there is less order in the lattice structure, so the atoms slide and reform bonds less well. All materials (brittle and ductile) experience an elastic phase, albeit potentially very short. At the end of the elastic phase, additional load induces intra-atomic movement or yield, which is when slip planes move against each other. Ductile metals have a limited elastic phase and a long yield phase before fracture.
What Are the Characteristics of a Ductile Metal?
The characteristics of a ductile metal are listed below:
- The high ductility metals can be drawn down to achieve astonishing length increases.
- Iron, in its cast and distributed carbon form, will fracture in brittle mode when the elastic limit is reached. However, ductile (or nodular) cast iron is highly ductile.
- Some ductility is the result of slip planes at an atomic level, typical of metals.
- Most ductile materials show a short and weak elastic phase, followed by a longer yield or ductile phase.
- All ductile materials will demonstrate brittle fracture at failure, eventually.
Where Are Ductile Metals Used?
Ductile metals are used in various manufacturing industries in which fine and complex forms are to be produced with relative ease. However, where ductility eases manufacture, it also increases toughness. Materials that can yield, will yield. Therefore when overloads are applied to components, they have a much higher functional survival rate if they remain ductile to some degree. In general, brittle materials are used where they will encounter little tensile or impact stress. Whenever loading patterns are complex and include tensile loads, vibration/fatigue loads, and impacts, ductile materials give better results.
What Are the Applications of Ductile Metals?
Listed below are some applications of ductile metals:
- Steel is typically worked in a mixture of ductile and malleable modes. Annealing and hot work are often used to increase ductility in rolling, bending, and forging.
- Aluminum is similarly worked, though the ductility is easier to maintain as it requires lower temperatures. Most aluminum rolling and shaping is performed cold and the work hardening that results strengthen/hardens the parts.
- All wire drawing exploits pure ductility. Some are performed cold (platinum, gold), some require annealing to achieve high extension (nickel, stainless steels), and some are performed hot (steels, stainless steels).
- Solders are highly ductile materials that make stress-tolerant joints between harder parts.
- Much jewelry work is undertaken cold and relies heavily on the ductility of the metals used.
Which Metal Has the Highest Ductility?
Gold has long held the title as the most ductile material, with a length of around 3,000 m per gram being commonly drawn. However, platinum drawn down using the Wollaston process (encased in silver as a stress shield/distributor) has been drawn to many times this length per gram.
How Is the Tensile Strength of Ductile Metals?
There is no direct interpretable connection between tensile strength and ductility. Tensile strength is often thought of as the stress or loading of a sample at fracture or structural failure, which is technically UTS (ultimate tensile strength). In functional part terms, there is an acceptable permanent offset limit that can be considered non-failure. Stress applied beyond the yield point that generates that limited extension is often referred to as the tensile strength of the material. Proof strength is perhaps more often used as a term for the safely useful tensile strength at or close to the elastic limit—i.e., the stress level from which a part or sample can recover fully and elastically.
How Is the Metal Ductility Being Measured?
There are two measures of ductility in common use. A clear comparison between these two measurements is not possible, however. In the first case, ductility is defined as the percentage reduction in the cross-sectional area resulting from the stress applied:
Ductility = (area after extension/area before extension) x 100%
Ductility is also defined as the percentage extension in length resulting from applied stress:
Ductility = (final sample length/original sample length) x 100%
In both cases, a standardized sample is progressively loaded in a tensile test machine until the yield point or onset of distortion is observed. This is the stress at which an observed change in the load-related extension rate occurs. Measurement of either cross-section or sample length is then recorded and the behavior in response to additional load is plotted as extension rate per unit load applied, generally up to failure/fracture.
How Can Ductility Be Improved in Metals?
Ductility in most metals can be improved by annealing. This process requires the metal to be uniformly heated to a temperature that is higher than the glass transition temperature but significantly lower than the softening temperature. This heating causes the crystal boundaries to dissolve, leaving the structure amorphous (when hot). As the metal cools, crystal growth restarts and the structure cools to a maximum general crystal size if the cooling is slow. Ductile behavior will begin to fracture these large crystals to a smaller size, generally hardening the material. This is commonly called work-hardening and it leads to brittle behavior and the end of ductility.
What Factors Affect the Ductility of Metals?
Metal ductility is primarily affected by two characteristics:
- A coarse crystal structure promotes ductility.
- A face-centered cubic crystal growth pattern is the most common in ductile metals.
Heat treatment can increase/restore ductility by annealing and remove/reduce it by hot quenching, which freezes a fine crystal structure into the material in some cases.
How Does Temperature Affect the Ductility of Metals?
For most metals, moderate temperature changes have a small influence on ductility. It is only as the glass transition temperature for the metal is reached that ductility will begin to increase, as crystal boundaries dissolve and the slip planes are able to move more freely. Some metals, such as lead and antimony, have glass transition temperatures at or below room temperature. They only develop a fixed crystalline structure when chilled. This renders them more ductile.
How Does the Ductile Metals are Used in Engineering?
Ductility is a property that is widely exploited in engineering materials, as an aid to easier processing of complex shapes. All drawn metal wires are the result of the exploitation of ductility, as the metal is pulled to reduce cross-section and increase length. Cold drawing, particularly of seamless stainless steel tubes is a common method to extend length in precise ways and induce work hardening. This is the exploitation of ductile behavior to the work hardening limit. In a less clear-cut way, the ability of metals to bend and flow into shapes, such as in forging or sheet metal pressing is the exploitation of ductility
Is Aluminum Ductile Metal?
Yes, aluminum in its pure form is a highly ductile metal and it can be drawn into very fine wires. As alloying agents increase, the hardness tends to rise and the ductility diminishes, but most aluminum alloys retail fairly high ductility.
Is Zinc Ductile Metal?
Yes, zinc is a highly ductile material when heated to a temperature between 110–150 °C and can be drawn into fine wires. Below this range it is crystalline and much less ductile; above this range, it begins to weaken and fractures more easily. For more information, see our guide on Brittle Failure.
Is Copper Highly Ductile Metal?
Yes, copper is very ductile and appears in any list of the most ductile metals.
What Is the Difference Between Ductility vs Malleability
Ductility is the ability of a material’s length to be extended under tensile load without fracture. Malleability, on the other hand, is a material’s ability to be formed and distorted under compressive (pressure or impact) loading without displaying internal failure or fracture.
Summary
This article presented ductile metals, explained what they are, and discussed the characteristics of each one To learn more about ductile metals, contact a Xometry representative.
Xometry provides a wide range of manufacturing capabilities and other value-added services for all of your prototyping and production needs. Visit our website to learn more or to request a free, no-obligation quote.
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.

