Manganese as a chemical element represents a critical component in modern metallurgy and industrial chemistry. Manganese refiners prioritize the extraction of the ore to satisfy global demand for high-strength infrastructure materials. Industrialists utilize the element to neutralize sulfur and oxygen during the smelting process. Diverse applications include battery manufacturing, fertilizer production, and glass coloring. Subsequent sections explore the atomic properties, chemical reactions, and specific roles of the substance in polymer science. Readers find detailed information regarding the atomic mass and periodic table classification of the metallic material. Specialized chapters discuss the impact of the element on stainless steel and carbon steel performance. Every application demonstrates the versatility of the transition metal in extreme industrial environments. Global economic growth remains linked to the availability of minerals containing the Manganese element for large-scale manufacturing projects.
What is Manganese as a Chemical Element?
Manganese, as a chemical element, is a gray-white transition metal that appears similar to iron but possesses a much harder and more brittle nature. Scientists categorize the substance within Group 7 of the periodic table due to its specific electron configuration. Manganese is essential as an alloying element in steel production for the production of low-cost stainless steel and high-impact alloys. Natural occurrences usually involve oxides or silicates found deep within the earth's crust. Refiners process the raw minerals to produce ferromanganese for large-scale metallurgical use. Manganese oxidizes in air and reacts slowly with water under certain conditions. Multiple oxidation states allow the element to participate in complex redox reactions within biological and industrial systems. Engineers rely on the material to increase the hardness and wear resistance of critical machine components. Global manufacturing depends significantly on manganese due to its critical role in steel production. Reliable performance in the furnace makes the metal a primary choice for heavy-duty industrial foundry work. Precise control over the chemical composition ensures the structural safety of modern bridges and high-rise buildings. The scientific community recognizes the substance as a vital building block for advanced material engineering. Foundries use controlled manganese content, typically via ferroalloys; ultra-high purity manganese is not generally required for structural steel.
What does Manganese Look Like as a Metal? Manganese looks like a silvery-gray metal with a hard and brittle texture. The surface appears lustrous when freshly polished but dulls quickly upon exposure to atmospheric oxygen. Polished specimens exhibit a metallic sheen comparable to traditional iron or nickel. Pure samples lack the ductility needed for direct structural use without alloying. Brittleness prevents the metal from being hammered into thin sheets or drawn into wires. Industrial users observe the material in a granulated or powdered form before the smelting process. Observation reveals a crystalline structure that shatters under high mechanical stress. Visual identification helps foundries rely on chemical analysis and standardized testing before it enters the molten iron.
How is Manganese Defined in Chemistry?
Manganese is defined in chemistry as a transition metal element known for having a wide range of oxidation states and powerful alloy-forming capabilities. Researchers observe the element participating in chemical bonds ranging from +2 to +7. The versatility enables the formation of various compounds like potassium permanganate and manganese dioxide. Chemists utilize the substance as a potent oxidizing agent in laboratory and industrial settings. Atomic structures facilitate the creation of strong magnetic properties when combined with other metals. Industrial chemistry processes rely on the element to catalyze reactions in the production of synthetic chemicals. The element serves as a vital micronutrient for enzyme activation in plant and animal metabolic pathways. Solubility in acidic solutions allows for the extraction and purification of the metal through hydrometallurgical methods. Knowledge of the chemical behavior helps engineers optimize the performance of the metal in modern manufacturing. Accurate chemical definitions support the development of high-capacity energy storage systems for electric vehicles. Every reaction involving the metal follows precise laws of stoichiometry and electron transfer. Scientific documentation identifies the metallic substance as an alloying or catalytic component in large-scale industrial chemistry. Reliability in the definitions ensures the consistent quality of products containing Manganese in various chemical states.
How Is Manganese Defined in Periodic Table? Manganese is defined in the periodic table as a transition metal located in Group 7 and Period 4. The placement reflects the electron configuration [Ar] 3d⁵ 4s², not only the 3d electrons. Chemists use the symbol Mn to represent the element in chemical equations and documentation. Placement among transition metals indicates variable oxidation states and the ability to form colored compounds; high melting point is not a defining criterion. Standard tables list the elements between chromium and iron on the horizontal row. The density is an experimentally measured physical property, approximately 7.43 grams per cubic centimeter. Atomic weight measurements provide a standard value for stoichiometric calculations in metallurgical processes.
Is Manganese an Element in the Periodic Table?
Yes, manganese is an element in the periodic table identified by the chemical symbol Mn. It occupies position 25 within Period 4 (first transition series). Periodic classification places the substance in Group 7 alongside technetium and rhenium. The location confirms its status as a d-block element with unique electronic properties. Scientists use the table to predict the reactivity and bonding patterns of the metal with other substances. Standard charts display the element as a solid at room temperature with a specific atomic mass. Education in material science starts with the recognition of the element in the transition series. Presence in the table ensures universal recognition in the scientific community.
What is the Atomic Number of Manganese?
The atomic number of manganese is 25. The integer represents the quantity of protons found within the nucleus of every atom belonging to the element. Protons determine the chemical identity and placement of the substance within the periodic system. Neutral atoms of the element contain 25 electrons to maintain electrical balance. Electron configurations follow the pattern of [Ar] 3d5 4s2 to explain magnetic and bonding behaviors. Atomic numbers remain constant regardless of the isotopic variation or physical state of the metal. Scientists utilize the number to distinguish the element from neighboring iron and chromium. Calculations involving the atomic number facilitate the understanding of ionization energy and electronegativity. Precise identification ensures that metallurgical recipes achieve the correct chemical balance during production. Modern physics relies on the integer to model the internal structure and stability of the atom. Consistent data support the widespread use of the transition metal in various technological applications. Atomic research confirms the importance of the proton count in defining the reactivity of the material. Precise measurements in the lab provide a solid foundation for industrial engineering. Every high-performance alloy depends on the unique characteristics of the Manganese atom with its 25 protons.
How Does the Atomic Number Identify the Manganese Element?
The atomic number identifies the manganese element by specifying the exact number of protons in its nucleus. Every element possesses a unique number of protons that serves as its chemical fingerprint. No other element in the universe contains exactly 25 protons within its atomic core. Accurate identification allows chemists to predict how the metal interacts with oxygen or sulfur. Scientists organize the periodic table by increasing atomic number to show recurring trends in material properties. The number 25 separates the substance from iron, which has 26, and chromium, which has 24. Atomic number identifies the element type, while purity is verified through compositional analysis e.g., spectroscopy. Researchers use spectroscopic techniques to count the electronic transitions associated with a specific number. Physical properties like density and melting point stem from the interaction of the 25 protons with the surrounding electrons. Reliable identification ensures the safety and quality of high-strength industrial components. Scientific standards rely on the atomic number to maintain the integrity of global chemical databases. Identification through atomic analysis supports the recovery of the metal from recycled scrap. Foundries adjust furnace conditions based on composition, temperature, and process parameters, not directly on proton count. Consistent results in the lab depend on the precise identification of Manganese through its atomic number.
Is the Atomic Number of Manganese 25?
Yes, the atomic number of manganese is 25. The value remains the defining characteristic of the element in all scientific literature. Periodic table entries list the number 25 directly above the chemical symbol Mn. Every atom of the metal must contain the specific number of protons to retain its identity. Measurement of the atomic number provides the basis for understanding the electronic structure of the d-block metals. Metallurgical calculations rely primarily on composition, phase behavior, and thermodynamics; atomic number is fundamental but not directly used in process calculations. Scientific consensus confirms the integer 25 as the permanent identifier for the substance.
What Are the Properties of Manganese?
The properties of Manganese are listed below.
- Hardness: Manganese represents a hard and brittle metal that shatters easily under high mechanical impact. The characteristic makes it unsuitable for pure structural use while enhancing the strength of metal alloys.
- Melting Point: The metal exhibits a high melting point of approximately 1246 degrees Celsius. Resistance to heat allows the element to maintain stability during high-temperature smelting processes.
- Oxidation States: Elements in the group display multiple oxidation states ranging from +2 to +7. Chemical versatility enables the formation of diverse compounds for battery and pigment manufacturing.
- Magnetism: Pure forms are paramagnetic, while certain alloys become strongly ferromagnetic. Magnetic properties vary depending on the temperature and the crystal lattice structure.
- Reactivity: Manganese oxidizes in air, but the oxide layer is not strongly protective and does not reliably prevent further oxidation. The metal reacts with dilute acids to release hydrogen gas and form various manganese salts.
- Density: Manganese has a density of 7.43 grams per cubic centimeter. The physical trait contributes to the weight and balance of high-strength Manganese alloyed steel products.
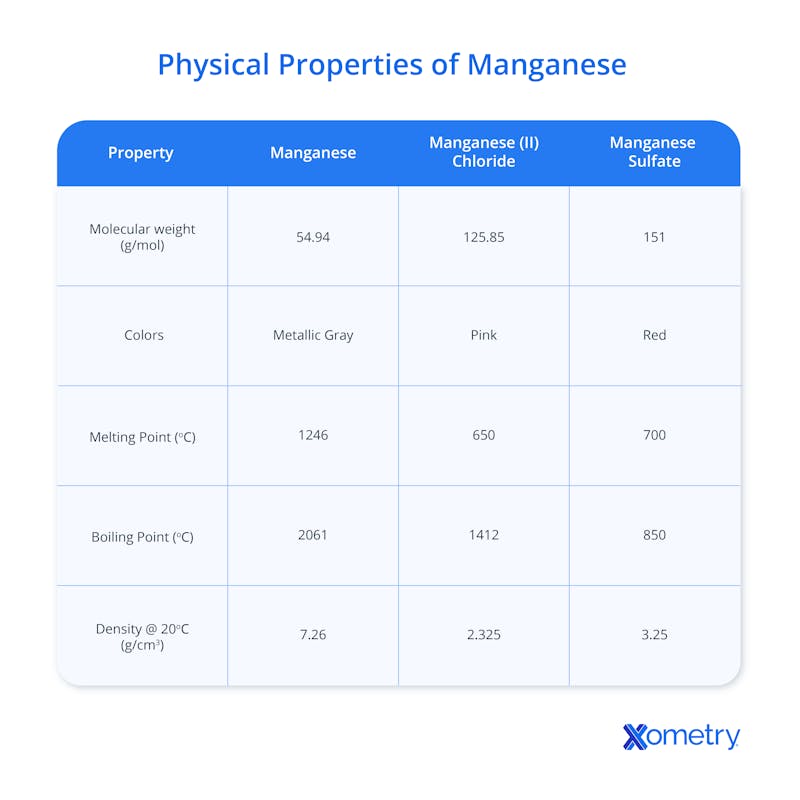
Which Physical Characteristics Define the Appearance of Manganese?
Physical characteristics defining the appearance of manganese include a hard, brittle, and silvery-gray metallic surface. Freshly fractured surfaces show a bright metallic luster that gradually fades upon exposure to moisture. The metal resembles iron in its coloration but lacks the ductility associated with most common steels. Granular textures appear in mineral samples found in deep-sea nodules or terrestrial mines. Brittle nature leads to a crystalline fracture pattern when the material shatters under pressure. The silvery hue shifts toward a brownish tint as the surface develops a thin layer of oxide. Solid forms maintain a fixed volume and shape at standard temperature and pressure conditions. Refined pellets appear uniform in color and texture for industrial processing. Crystalline structures are visible under magnification to reveal the internal arrangement of the atoms. Visual identification may assist initial sorting, but accurate separation requires chemical analysis and mineral processing techniques. Producers rely on chemical composition analysis and quality testing rather than appearance alone. Metallic samples retain a dense and heavy feel when held in the hand. Polishing the surface restores the original brilliance of the transition metal temporarily. Ore grading is determined through chemical assays and analytical techniques.
Is Manganese Naturally a Solid Metal?
Yes, manganese is naturally a solid metal at room temperature and standard atmospheric pressure. It exists in a dense crystalline state with a high degree of structural rigidity. The transition metal remains in the solid phase until temperatures exceed 1246 degrees Celsius. Most natural deposits occur as solid oxide minerals like pyrolusite within the earth's crust. Extraction processes yield a Solid Metal that refiners utilize for diverse industrial applications. Stability in the solid phase ensures easy transportation and storage for global manufacturing.
What Are the Chemical Properties of Manganese?
The chemical properties of manganese are listed below.
- Manganese exhibits multiple oxidation states from +2 to +7 in various chemical environments.
- The metal reacts very slowly with water under normal conditions; the reaction with water is limited.
- Exposure to air causes the formation of a thin oxide layer that protects the interior metal.
- Reaction with dilute acids produces manganese(II) salts and hydrogen gas.
- The element functions as a powerful reducing agent in high-temperature metallurgical reactions.
- Manganese dioxide acts as a catalyst in the decomposition of hydrogen peroxide into water and oxygen.
- Chemists utilize the high reactivity of Manganese to synthesize complex organic and inorganic compounds.
How Does Manganese React with Other Elements in Chemical Reactions?
Manganese reacts with other elements in chemical reactions by forming a variety of compounds with oxygen, sulfur, and the halogens. Oxidation in air can produce a mixture of oxides (e.g., MnO, Mn₂O₃, Mn₃O₄), not exclusively manganese(II) oxide. Halogen gases like chlorine react with the heated metal to produce manganese chlorides. Combustion in pure oxygen results in the formation of trimanganese tetroxide under specific thermal conditions. The metal combines with sulfur at high temperatures to produce manganese sulfide. Multiple valence electrons allow the substance to bond with diverse ligands in coordination chemistry. Industrial processes utilize the reactions to purify the metal and create useful chemical reagents. High reactivity toward impurities like sulfur makes the element indispensable in the refinement of molten iron. Chemical bonds form, forming compounds (e.g., MnS) and modifying microstructure, during the processes, improving the structural integrity of the final product. Understanding the interactions helps scientists develop more efficient catalysts for the chemical industry. Refiners monitor the reaction rates to control the quality of large-scale industrial outputs. Chemical stability of the resulting compounds determines their use in pigments and agricultural products. Every reaction follows the principles of electron sharing and transfer between atoms. Industrial safety protocols govern the handling of reactive Manganese dust in the workplace.
Can Manganese React with Oxygen to Form Compounds?
Yes, manganese reacts with oxygen to form various manganese oxides, such as manganese dioxide and trimanganese tetroxide. The reaction starts at room temperature with the formation of a dull surface coating on the metal. Higher temperatures accelerate the oxidation process during industrial smelting or welding. The oxides serve as critical components in dry cell batteries and ceramic pigments. Catalytic properties of the oxides support the synthesis of various industrial chemicals. The ability to form oxygen compounds demonstrates the high chemical activity of the metal.
Manganese functions primarily as a metallurgical process element that governs impurity control and phase stability in steel systems. Its most critical role is in deoxidation and desulfurization, where it preferentially reacts with oxygen and sulfur to form stable oxides and manganese sulfides, preventing the formation of brittle iron sulfide phases that would otherwise cause hot shortness. In addition, manganese increases hardenability by lowering the critical cooling rate required for martensitic transformation, enabling deeper and more uniform hardness profiles in heat-treated steels. It also contributes to solid solution strengthening and grain refinement, improving toughness and wear resistance under cyclic and impact loading conditions. While it does not provide standalone structural utility due to its brittleness, its alloying effect is essential for producing high-performance carbon and stainless steels. Beyond metallurgy, its multiple oxidation states enable catalytic functionality in polymerization and chemical synthesis processes. In practice, manganese is not selected for direct application, but for its ability to control microstructure, purity, and performance of engineered material systems.
How is the Atomic Mass of Manganese Measured in Chemistry?
The atomic mass of manganese is measured in chemistry through the use of mass spectrometry and isotopic analysis of natural samples. Mass spectrometers separate individual atoms based on their mass-to-charge ratio in a vacuum. Detectors count the number of atoms for each isotope to determine their relative abundance in the sample. Scientists compare the results to standard reference materials to ensure high accuracy. Mathematical averages weigh the mass of each isotope according to its frequency in nature. Isotopic analysis helps researchers identify the origin of mineral deposits in geological studies. Precise measurements allow for the calculation of molar masses used in industrial chemical synthesis. Laboratories refine the values as better technology becomes available for atomic detection. The process ensures that stoichiometric ratios in metallurgy are controlled by composition and process control across the global supply chain. Constant monitoring of isotopic ratios reveals changes in environmental conditions over geological time. Accurate data regarding the mass facilitates the production of high-quality industrial components. Scientific equipment requires calibration using known standards to maintain measurement integrity. International organizations publish updated mass values to guide research and development efforts. Foundries utilize the figures to achieve precise chemical balances in the production of Manganese alloyed products.
Is the Atomic Mass of Manganese Approximately 54.94?
Yes, the atomic mass of manganese is approximately 54.94 atomic mass units according to standard scientific measurements. The value appears in all modern periodic tables and chemical reference books. Molar calculations in chemistry rely on the specific figure to achieve accurate reaction results. The number represents the average weight of the protons, neutrons, and nuclear binding effects found in the nucleus. Metallurgists use the mass to balance chemical equations during the steelmaking process. Consistency in the measurement ensures the reliability of international manufacturing standards.
What Are the Main Uses of Manganese?
The main uses of Manganese are listed below.
- Steel production: Manganese acts as a deoxidizer and desulfurizer to improve the strength and workability of structural steel. The application consumes the vast majority of the global production of the metal.
- Alloy manufacturing: The element combines with aluminum and copper to create durable, corrosion-resistant metals for marine and aerospace use. Alloying can increase hardness, but it may also change density and weight depending on composition.
- Battery production: Manganese dioxide serves as a cathode material in alkaline and lithium-ion batteries for electronic devices. High purity levels are required to ensure the efficiency and safety of power cells.
- Fertilizers and plant nutrients: Manganese acts as a vital micronutrient that supports photosynthesis and enzyme activation in various crops. Agriculture industries utilize the metal to prevent yellowing and stunted growth in plants.
- Glass and ceramics production: Compounds of the metal neutralize the green tint caused by iron impurities in glass manufacturing. Higher concentrations produce purple or brown colors in decorative ceramic glazes.
- Chemical manufacturing: Manganese salts function as catalysts in the production of synthetic fabrics and organic chemicals. Industrial chemistry relies on catalysts to accelerate reaction rates and improve yields.
- Water treatment: The element helps remove iron and hydrogen sulfide from drinking water supplies through oxidation. Treatment plants utilize specialized filters to capture the resulting solid particles.
- Pigments and dyes: Oxides of the metal provide black, brown, and purple pigments for paints and industrial coatings. Stability in the pigments ensures long-lasting color for exterior and interior surfaces.
- Animal feed additives: Livestock diets include small amounts of the element to support bone development and reproductive health. Producers add the mineral to ensure the overall well-being of the animals.
- Electronics manufacturing: Magnetic alloys containing the element are used in the production of sensors and memory devices. Electronic components benefit from the specific thermal and magnetic properties of Manganese in various circuit designs.
Why is Manganese Important for Industrial Applications?
Manganese is important for industrial applications because it improves the mechanical properties of metals and functions as an efficient catalyst in chemical synthesis. Manganese is highly effective and widely used for desulfurization, but other elements (e.g., calcium, magnesium) are also used depending on the process. The substance improves the hardness and toughness of steel needed for high-impact machinery. Industrial catalysts containing the element accelerate the production of essential polymers and synthetic fibers. Resistance to corrosion makes the metal indispensable for marine hardware and specialized construction materials. Energy sectors rely on the element for high-capacity battery storage systems and portable electronics. Agricultural productivity increases through the use of the metal as a critical micronutrient in soil amendments. Glass manufacturers use the material to achieve high clarity in optical and consumer products. The versatile nature of the element allows it to bridge the gap between heavy metallurgy and fine chemical engineering. Modern infrastructure depends on the consistent performance of the metal in extreme environments. Global economic growth remains linked to the availability of the metal for large-scale manufacturing projects. Steelmakers prioritize the sourcing of high-grade ore to maintain the quality of their industrial output. Versatility across different sectors makes the metal a primary commodity in global trade. Manufacturing efficiency improves through the use of specialized Manganese catalysts in the production of complex resins.
Is Manganese Widely Used in Industrial Manufacturing?
Yes, manganese is widely used in industrial manufacturing with a primary focus on the steel and alloy industries. It serves as an essential component for the production of durable construction materials and automotive parts. Global consumption of the metal reaches millions of tons annually to satisfy the demands of urbanization. Secondary applications in battery technology and chemical processing continue to expand in modern markets. Presence in diverse products confirms its role as a versatile industrial tool. Manufacturing sectors rely on the material to improve product quality and performance.
How Does Manganese Compare to Other Metals?
Manganese compares to other metals by offering a much higher degree of hardness and brittleness while providing superior alloying benefits for iron. The most common metals, like copper or aluminum, exhibit higher ductility and electrical conductivity. The element resembles iron in color and density, but does not serve as a standalone structural material. Unlike gold or platinum, the metal oxidizes readily when exposed to moisture and oxygen. Manganese is widely used for sulfur control, but other elements (e.g., calcium, magnesium) also perform this function. High-capacity battery applications place it alongside cobalt and nickel as a critical energy material. The substance offers a more affordable alternative to expensive, rare earth elements in certain magnetic alloys. Metallurgical stability allows the metal to perform well under extreme temperatures where others might soften. Researchers study the element to find new ways of replacing costly metals in industrial catalysts. Unique electronic configurations provide a wider range of oxidation states than many neighboring elements in the d-block. Diverse properties ensure that the substance remains a unique and valuable asset in the periodic table. Structural engineers compare the hardness of the metal to that of hardened steel when evaluating alloy performance. Every metallurgical comparison highlights the role of the metal in enhancing the toughness of iron. Foundries utilize the unique reactivity of Manganese to refine molten metals in the production of heavy machinery.
What Differences Exist Between Manganese and Iron?
The differences between Manganese and Iron are listed below.
- Brittleness: Manganese is extremely brittle and shatters upon mechanical impact, while iron exhibits higher ductility and malleability. The difference limits the use of the pure metal to alloying rather than direct construction.
- Oxidation States: Elements in the manganese group reach higher oxidation states like +7 compared to the typical +2 and +3 seen in iron chemistry. Greater chemical versatility allows for a wider range of compounds and catalytic applications.
- Magnetic Behavior: Pure manganese is paramagnetic, while iron is famously ferromagnetic at room temperature. Magnetic properties of the metals influence their use in electronic and structural engineering.
- Melting Points: The melting point of manganese is approximately 1246 degrees Celsius, which is lower than the 1538 degrees Celsius required for iron. Thermal differences affect the behavior of the metals during the casting and smelting process.
- Chemical Reactivity: Manganese reacts readily with dilute acids, but its reaction with water is limited; comparison with iron depends on conditions. Surface oxidation occurs on both metals, but results in different chemical structures and appearances for Manganese and iron.
Is Manganese Harder Than Pure Iron?
No, Manganese is not reliably harder than pure iron; hardness comparison depends on the measurement method. The increased hardness contributes to the strength of steel alloys used in high-wear applications. Brittleness accompanies the hardness, preventing the metal from being easily forged into complex shapes. Pure iron remains softer and more easily deformed under mechanical pressure. Industrialists value the element for its ability to impart hardness to iron during the alloying process. The difference in physical properties ensures each metal serves a distinct role in metallurgy.
How is Manganese Used in Plastic Materials and Polymer Manufacturing?
Manganese is used in plastic materials and polymer manufacturing as a catalyst and stabilizer to regulate chemical reaction rates. Compounds like manganese acetate facilitate the production of various polyester resins used in fabrics and packaging. The element helps control the polymerization process to ensure consistent material density and strength. Stabilizing agents containing the metal prevent the degradation of plastics when exposed to heat and sunlight. Industrialists utilize the compounds to improve the durability and longevity of consumer products. Catalytic systems involving the metal offer a cost-effective alternative to precious metal catalysts. Presence in the manufacturing process influences the molecular weight and chain structure of the resulting polymers. Researchers investigate new ways to use the metal in the synthesis of specialty engineering plastics. The applications expand the utility of the element beyond traditional metallurgical sectors. Consistent performance in chemical reactors ensures the quality of high-volume plastic production. Quality control teams monitor the concentration of the metallic additives to maintain safety standards. The role of the metal in catalysis supports the transition toward more efficient manufacturing methods. Durable plastic components for the automotive industry benefit from the presence of specialized stabilizers and fillers. Modern industries rely on the catalytic efficiency of Plastic Materials and Polymer additives during the synthesis phase.
What Role Does Manganese Play in the Production of High Density Polyethylene (HDPE)?
Manganese plays a role in the production of high density polyethylene by serving as a component in certain catalytic systems that facilitate ethylene polymerization. Specialized catalysts containing the element help control the formation of long, unbranched polymer chains. High density levels result from the precise arrangement of the molecular structures during the reaction. The presence of the metal influences the reaction speed and the overall efficiency of the chemical plant. Manufacturers adjust the catalyst concentration to achieve the desired stiffness and impact resistance in the plastic. The catalysts help produce a material that is ideal for heavy-duty containers and industrial pipes. High purity levels are maintained to ensure the plastic remains safe for food and medical applications. Industrial chemistry continues to refine the catalytic roles to reduce production costs and environmental impact. Strength and durability of the finished product depend on the quality of the initial polymerization. Modern packaging solutions rely on the structural integrity provided by the metal-assisted reactions. The element helps regulate the molecular weight distribution within the plastic resin. Large-scale production facilities utilize the catalysts to maintain high output levels. Reliability in the processes ensures the consistent supply of High Density Polyethylene (HDPE) for the global market.
Can Manganese Compounds Be Used as Catalysts in High Density Polyethylene (HDPE) Production?
Yes, certain manganese compounds can function as catalysts in the polymerization processes used to create high density polyethylene. The compounds assist in the conversion of ethylene gas into solid plastic structures with high molecular weight. Catalytic activity helps regulate the density and tensile strength of the resulting material. Manufacturers choose the catalysts to optimize the production efficiency of large-scale industrial plants. Accurate control of the catalyst ensures the production of high-quality plastic goods.
How Does Manganese Relate to Engineering Plastics Used in Manufacturing?
Manganese relates to engineering plastics used in manufacturing through limited and specialized functions rather than broad performance improvement. Compounds containing the metal do not serve as the primary method for preventing oxidative degradation during high-temperature processing. Oxidative degradation in polymers is controlled by antioxidants and stabilizers that interrupt free radical reactions and protect polymer chains. The plastics must withstand extreme mechanical stress and environmental exposure in automotive and aerospace applications. The element serves as a catalyst in the synthesis of specialty polyamides and polycarbonates. Industrialists utilize the materials to replace heavier metal components in complex machinery. Stabilizing agents ensure the plastics maintain their color and structural integrity over long service periods. Catalytic roles support the efficient production of resins with precise molecular architectures. Advancements in polymer science continue to discover new ways to utilize the element in material manufacturing. High-performance plastics benefit from the consistent quality provided by the metallic additives. Engineers rely on the improved properties of the polymers to design more durable mechanical parts. Versatility in the chemical roles of the metal expands its presence in modern engineering labs. Reliability in the stabilizing process ensures the longevity of plastic components in harsh conditions. Designers rely on the improved properties of Injection Molding materials to create more durable and efficient products.
How Are Manganese Compounds Applied in Processes Involving Acetal (POM)?
Manganese catalysts do not influence acetal (POM) polymer production in standard industrial practice. Acetal polymerization depends on cationic initiation systems, where compounds such as boron trifluoride activate formaldehyde or trioxane to form polyoxymethylene chains through a controlled ionic mechanism. Cationic initiators provide the required reactivity and chain propagation behavior that defines POM structure, molecular weight, and stability.
POM production follows a defined sequence that includes polymerization, end-group stabilization, and compounding with antioxidants and thermal stabilizers. Each stage relies on chemistry aligned with cationic processes rather than transition metal catalysis. Manganese-based catalysts operate through redox or coordination pathways that do not match the reaction mechanism required for acetal formation. Industrial systems exclude manganese compounds because they do not support efficient chain growth or stability control in Acetal (POM) synthesis.
Can Manganese Catalysts Influence Acetal (POM) Polymer Production?
No, manganese catalysts do not influence acetal (POM) polymer production in standard industrial practice. Acetal polymerization depends on cationic initiation systems, where compounds such as boron trifluoride activate formaldehyde or trioxane to form polyoxymethylene chains through a controlled ionic mechanism. Cationic initiators provide the required reactivity and chain propagation behavior that defines POM structure, molecular weight, and stability.
POM production follows a defined sequence that includes polymerization, end-group stabilization, and compounding with antioxidants and thermal stabilizers. Each stage relies on chemistry aligned with cationic processes rather than transition metal catalysis. Manganese-based catalysts operate through redox or coordination pathways that do not match the reaction mechanism required for acetal formation. Industrial systems exclude manganese compounds because they do not support efficient chain growth or stability control in POM synthesis.
How is Manganese Connected to Flexible Polymer Materials?
Manganese connects to flexible polymer materials only in limited and specialized contexts rather than as a standard additive. Flexible polymers rely on plasticizers, stabilizers, and polymer structure to achieve softness, elasticity, and durability. Manganese does not function as a primary component for improving flexibility, thermal stability, or chemical resistance in these materials.
Manganese compounds appear in niche roles such as catalysts in certain polymer-related reactions or as pigments and driers in coatings applied to flexible substrates. Flexible polymer systems such as PVC, TPU, and elastomers depend on additives (plasticizers, antioxidants, UV stabilizers) that directly control flexibility and long-term performance. Manganese does not replace or perform the functions of these additives, which limits its relevance in mainstream flexible Polymer Materials used in everyday products.
What Role Can Manganese Play in Processing Thermoplastic Elastomers (TPE)?
Manganese does not play a standard role in processing thermoplastic elastomers (TPE). Thermoplastic elastomer processing relies on additive systems designed for stability, durability, and performance control rather than transition metal compounds. Manufacturers use antioxidants, UV stabilizers, and processing aids to protect polymer chains from heat, oxygen exposure, and environmental stress during extrusion, injection molding, and compounding stages.
Thermoplastic elastomer formulations depend on base polymers (styrenic block copolymers, polyolefin blends, TPU) combined with additives that control oxidation, light degradation, and flow behavior. Stabilization systems include hindered phenols, phosphites, and UV absorbers that maintain elasticity and mechanical strength under repeated thermal cycles. Manganese compounds do not align with these functions and do not appear as standard catalysts or stabilizers in Thermoplastic Elastomers (TPE) production. Industrial practice limits manganese use to niche or unrelated applications rather than core thermoplastic elastomer processing.
Can Manganese Compounds Be Used in Thermoplastic Elastomer (TPE) Processing?
No, manganese compounds are not commonly used in thermoplastic elastomer (TPE) processing or stabilization. TPE manufacturing depends on polymer design and conventional additive systems rather than transition metal compounds. Standard formulations include antioxidants and UV stabilizers that protect polymer chains from heat, oxygen, and light during extrusion and molding operations.
Thermoplastic elastomers derive performance from base polymer structures (styrenic block copolymers, thermoplastic polyurethanes, polyolefin blends) combined with stabilizers that maintain elasticity and durability. Additives such as hindered phenols and phosphites control oxidative degradation, while UV absorbers protect against sunlight exposure. Manganese compounds do not provide these functions and do not appear in standard industrial formulations for TPE production.
Why is Manganese Important in Stainless Steel Manufacturing?
Manganese is important in stainless steel manufacturing because it serves as an essential alloying element that improves hardness and resistance to wear. It acts as a deoxidizer to remove oxygen and other impurities from the molten metal during the smelting process. The element combines with sulfur to form manganese sulfide, preventing the formation of brittle iron sulfides. Many grades of stainless steel utilize the metal as a lower-cost alternative to nickel for stabilizing the austenitic structure. The substitution allows for the production of high-performance steel at a more affordable price point. The substance improves the workability of the alloy during hot rolling and forging operations. High-strength stainless steels used in construction and transportation rely on the metal for structural integrity. Industrialists monitor the concentration of the metal to ensure the steel meets international quality standards. The versatile nature of the element makes it indispensable for creating durable and reliable metal products. Construction firms specify the overall alloy compositions and standards for projects requiring long-term structural safety. Every high-rise building incorporates metallic components in the structural frame. The role of the metal in deoxidation ensures a dense and defect-free final product. Global steel markets depend on the consistent availability of Stainless Steel components for modern infrastructure.
How Does Manganese Improve the Properties of Stainless Steel (SS)?
Manganese improves the properties of stainless steel by increasing its tensile strength and resistance to mechanical impact. The element facilitates the stabilization of the austenite phase, which provides the steel with excellent toughness at low temperatures. Alloying with the metal improves the hardness of the surface, making it more resistant to abrasive wear. It contributes to the overall corrosion resistance of the material in certain chemical environments. The presence of the metal ensures the steel stays easily welded and machined without losing its structural integrity. Deoxidation during the melting phase results in a cleaner metal with fewer internal defects. The improvements make the steel ideal for use in heavy-duty machinery and industrial equipment. Manufacturers adjust the alloy composition to achieve the specific performance characteristics required for each application. The metal acts as a critical building block for high-performance materials in the aerospace and automotive industries. Strength and durability of the finished product depend on the precise control of the alloying process. Reliable metallurgical practices support the production of the alloy for diverse global markets. Heavy-duty fasteners and valves rely on the mechanical improvements provided by the metal. Modern industrial systems benefit from the longevity of the high-strength materials. Reliable metallurgical practices support the production of high-quality Stainless Steel for diverse global markets.
Is Manganese Commonly Added to Stainless Steel Alloys?
Yes, manganese is commonly added to stainless steel alloys to improve their structural strength and industrial usefulness. It serves as a vital component in many 200-series and 300-series stainless steels. The element provides a cost-effective way to improve the hardness and durability of the metal. Metallurgists utilize the metal to replace a portion of the nickel content without compromising performance. Presence in meeting performance demands depends on the overall alloy composition and processing, ensuring the steel meets the rigorous demands of modern construction and manufacturing.
How is Manganese Used in Carbon Steel Production?
Manganese is used in carbon steel production as a primary deoxidizer and desulfurizer during the initial smelting process. It reacts with oxygen and sulfur impurities to form solid slag that can easily be removed from the molten iron. The purification process prevents the formation of weak spots and cracks in the finished steel. The element acts as an alloying agent to increase the hardness and tensile strength of the material. Carbon steel containing the metal exhibits improved workability during the rolling and forging stages. Strength and durability remain essential for structural beams, pipes, and automotive components. The substance helps refine the grain structure of the metal to improve its overall toughness. Many industrial standards require a minimum concentration of the metal to ensure the safety and reliability of the steel. Industrialists choose the element because it offers superior performance at a lower cost than many alternatives. Consistent quality in the production cycle ensures the steel remains compatible with high-speed manufacturing lines. Foundries prioritize the use of the metal to achieve high-density castings for heavy machinery. Reliability in structural engineering depends on the consistent behavior of the metal alloys. Large-scale infrastructure projects rely on the strength of the purified steel products. Precise metallurgical control supports the widespread use of Carbon Steel in global infrastructure projects.
What Metallurgical Function Does Manganese Serve in Carbon Steel (CS)?
Manganese serves several metallurgical functions in carbon steel, including the prevention of hot shortness caused by sulfur impurities. It combines with sulfur to form stable manganese sulfide particles that do not melt at the temperatures used for hot working. The function allows the steel to be rolled and forged without tearing or breaking during production. The element increases the hardenability of the metal, allowing it to reach greater depth during heat treatment processes. It improves the response of the steel to quenching and tempering cycles used to optimize strength. Deoxidation reduces oxygen-related defects and inclusions, but density and porosity are primarily controlled by solidification and processing conditions. The deoxidizing role ensures a high-density material with minimal porosity in the internal structure. The functions make the element indispensable for the production of reliable structural and automotive steels. Engineers utilize the specific properties of the alloy to meet the demanding requirements of modern machinery. Strength and toughness of the metal are improved through the precise addition of the element. Structural stability in bridge construction depends on overall steel composition, design, and processing; manganese contributes but is not solely responsible. Metallurgical analysis confirms the importance of the element in refining the metal grain. Every high-strength structural bolt requires the presence of the alloying components. Reliable metallurgical data support the use of Carbon Steel in heavy-duty industrial applications.
Does Manganese Increase the Strength of Carbon Steel?
Yes, manganese increases the strength of carbon steel by improving its hardness and tensile resistance. The element acts as a critical alloying agent that improves the mechanical properties of the base iron. Alloying allows the steel to withstand higher loads and mechanical stress without failure. Hardness improves through the refinement, solid solution strengthening, and increased hardenability. Industrialists rely on the strength to produce durable construction materials and machine parts. Consistent use of the metal ensures the reliability of the finished product.
How is Manganese Related to Advanced Injection Molding Techniques?
Manganese is related to advanced injection molding techniques through its presence in high-performance steel alloys used for tooling and molds. The alloys must withstand the extreme heat and pressure associated with modern molding cycles. The element increases the hardness and wear resistance of the mold surfaces to prevent erosion from abrasive resins. Tooling alloys containing the metal provide superior dimensional stability over long production runs. Advanced techniques like gas-assisted molding benefit from the use of durable and reliable mold materials. Resistance to mechanical impact ensures the molds remain functional even under high-speed automated operation. Manufacturers choose the alloys to reduce the downtime and maintenance costs associated with mold wear. Precise control over the material properties ensures the production of high-quality plastic components. Reliability in the processes supports the growth of the manufacturing industry. High-precision medical components depend on the durability of the metal-alloyed molds. Modern factories utilize advanced materials to achieve higher production efficiency. Reliability in the processes supports the growth of the Injection Molding industry.
How Can Gas Assisted Injection Molding Benefit from Manganese Alloyed Tooling?
Gas assisted injection molding can benefit from manganese alloyed tooling by utilizing the increased hardness and wear resistance of the mold surfaces. The technique involves the injection of pressurized gas into the molten resin to create hollow, lightweight parts. The mold must maintain high structural integrity to handle the localized pressure changes during the gas injection stage. Alloying with the metal improves the fatigue resistance of the steel, extending the service life of complex molds. Higher surface hardness prevents the formation of scratches and pits that ruin the aesthetic finish of the plastic part. Improved thermal stability ensures the mold remains dimensionally accurate throughout repeated heating and cooling cycles. Manufacturers achieve higher production efficiency by reducing the frequency of mold repairs and replacements. Knowledge of metallurgical properties helps engineers design more durable and reliable tooling for advanced molding processes. Consistency in the tool quality leads to superior part performance and reduced waste. Reliability in the mold structure ensures safe operation under high gas pressure. Designers utilize high-strength materials to create more complex and efficient plastic goods. Reliable performance is a hallmark of Injection Molding systems utilizing high-strength alloys.
Is Manganese Used in Tooling Materials for Gas Assisted Injection Molding?
Yes, manganese is present in the steel alloys used for tooling materials in gas assisted injection molding. The element provides the necessary hardness and toughness for molds subjected to high-pressure cycles. Alloying improves the resistance of the mold surfaces to wear and mechanical stress. Industrialists choose the alloys to ensure the longevity and reliability of their manufacturing equipment. High-performance steel containing the metal supports the production of complex and high-quality plastic components. Consistent material properties are essential for process stability and efficiency.
What Are the Advantages of Using Manganese?
The advantages of using Manganese are listed below.
- Desulfurization: Manganese effectively removes sulfur from molten iron to prevent the formation of brittle phases. The advantage ensures that steel remains workable and strong during industrial forging.
- Deoxidation: The element reacts with oxygen impurities to create a cleaner and more stable metal alloy. Reducing oxygen content prevents the formation of voids and defects in the internal structure.
- Hardenability: Alloying with the metal increases the depth to which the steel stays hardened through heat treatment. The trait allows for the production of heavy-duty structural components with uniform strength.
- Cost-Effectiveness: Manganese serves as a relatively affordable alloying agent compared to nickel or cobalt. Manufacturers achieve high mechanical performance without increasing production costs.
- Catalytic Activity: Compounds of the metal serve as efficient catalysts in the synthesis of polymers and chemicals. Catalysis improves the speed and yield of various industrial manufacturing processes.
- Thermal Stability: Manganese contributes to mechanical properties, but high-temperature stability in alloys depends more on elements like chromium, molybdenum, and nickel. Resistance to thermal softening supports the use of the metal in Manganese steel for heavy industrial applications.
How Xometry Can Help
While we don’t offer manganese in it’s pure form, as discussed it’s a crucial element in many of the steel and aluminum alloys Xometry offers. We offer a range of manufacturing processes such as CNC machining, sheet metal fabrication, and sheet cutting, all of which can work with alloys enhanced by manganese. Best of all, projects that require these processes can all be instantly quoted through our website. You can get started by uploading your 3D CAD files to the Xometry Instant Quoting Engine® and get instant pricing and lead times today!
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.


